| Issue |
J Oral Med Oral Surg
Volume 32, Number 1, 2026
|
|
|---|---|---|
| Article Number | 7 | |
| Number of page(s) | 12 | |
| DOI | https://doi.org/10.1051/mbcb/2025042 | |
| Published online | 10 April 2026 | |
Systematic Review
Etiology, clinical features, and management strategies in orofacial rhinosporidiosis: a systematic review
1
Oral Medicine and Radiology, Meenakshi Ammal Dental College, Meenakshi Academy of Higher Education and Research, Vembuliamman Koil Street, West KK Nagar, Chennai, India
2
Internal Medicine, Meenakshi Academy of Higher Education and Research, Vembuliamman Koil Street, West KK Nagar, Chennai, India
3
Department of Research, Meenakshi Academy of Higher Education and Research, Vembuliamman Koil Street, West KK Nagar, Chennai, India
* Correspondence: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
3
October
2025
Accepted:
15
December
2025
Abstract
Background: Rhinosporidiosis, caused by Rhinosporidium seeberi, mainly affects the nasal mucosa but can also involve orofacial sites such as the oral cavity, maxillary sinus, and buccal mucosa, complicating diagnosis and treatment. Endemic in India, Sri Lanka, and parts of South America, infection is linked to stagnant water exposure, yet orofacial cases remain underreported with limited management guidance. Methodology: This systematic review followed PRISMA guidelines to ensure transparency. Comprehensive searches of PubMed, Scopus, Web of Science (2012–2025) identified 238 records, with 16 studies included after screening. English-language case reports and series reporting clinical, diagnostic, or therapeutic data on orofacial rhinosporidiosis were considered eligible. Data was extracted using standardized forms, and study quality was assessed with the Joanna Briggs Institute checklist, evaluating selection, reporting, and outcome bias. Results: Analysis of reported cases shows that orofacial rhinosporidiosis primarily affects young to middle-aged men, with females rarely affected. Patient ages ranged from 10 to 65 yr. Frequently involved sites include the nasal cavity, maxillary sinus, and nasopharynx, with occasional lesions in the oral cavity, oropharynx, buccal mucosa, and palate; disseminated involvement of subcutaneous tissues or lungs is rare. Lesions are polypoid, reddish, friable, and “strawberry-like”, with symptom duration spanning weeks to 20 yr. Diagnosis is confirmed via histopathology, with imaging used selectively. Surgical excision with electrocauterization, often supplemented by Dapsone, yields favorable outcomes. Follow-up ranged 3–36 months, with minimal complications, though rare hemolytic anemia was reported. Conclusion: Orofacial rhinosporidiosis primarily affects young to middle-aged men, presenting as polypoid, “strawberry-like” lesions that require histopathological confirmation for diagnosis. Surgical excision with electrocauterization, supplemented by Dapsone when appropriate, provides effective management, though long-term follow-up is essential to monitor for recurrence.
Key words: Chronic granulomatous disease / mucosal involvement / nasopharynx / nodular swelling / orofacial infection / polypoidal lesion / Rhinosporidium seeberi / rhinosporidiosis / sporangia / vascular proliferation
© The authors, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Rhinosporidiosis is a chronic granulomatous disease caused by Rhinosporidium seeberi, an aquatic protistan parasite that primarily affects mucous membranes of the nasal cavity and nasopharynx. However, several orofacial sites including the oral cavity, maxillary sinus, and buccal mucosa are often involved which presents with unique diagnostic and therapeutic challenges [1–4]. The disease is endemic in certain tropical and subtropical regions, specifically in India, Sri Lanka, and parts of South America, where environmental exposure to stagnant water bodies plays a significant role in this transmission [5–8].
The prevalence and occasional atypical presentations revealed from epidemiological data, which is gathered from rural and endemic regions, has emphasized the public health relevance of this disease [9–12]. Moreover, there are reports of cases of rhinosporidiosis occurring in different anatomical sites apart from the classical nasal and nasopharyngeal sites. Thus, this signifies the importance of awareness among clinicians to consider it a possible differential diagnosis of orofacial masses, particularly in endemic areas [13–17]. However, the etiology, clinical presentation, and optimal management strategies for orofacial manifestations remain unknown in the scientific literature when compared to nasal rhinosporidiosis. Some systematic reviews have summarized such cases geographically or focused on classical nasal presentations [6,7]. However, there is a gap in a comprehensive synthesis that addresses orofacial rhinosporidiosis, its pathogenesis, diverse clinical presentations, diagnostic challenges, and therapeutic algorithms. There is also limited agreement on management strategies for atypical or disseminated cases, and the role of adjunctive pharmacotherapy remains controversial [18–21]. Moreover, novel surgical innovations and diagnostic modalities have not been systematically reviewed in the context of orofacial involvement.
Clinically, orofacial rhinosporidiosis characteristically manifests as polypoidal masses or nodular swellings, which may mimic other benign or malignant lesions, complicating early diagnosis [14,22]. Uncommon presentations such as large oral cavity masses and facial swellings have been reported, highlighting the variability of clinical features and the need for accurate clinical acumen [2,3,13]. Imaging studies often produce non-specific findings which further leads to the diagnostic dilemma, especially when the lesion involves uncommon sites like the maxillary sinus or causes orbital complications such as proptosis [4,23,24]. Histopathological examination remains the gold standard for definitive diagnosis, demonstrating characteristic sporangia filled with endospores within a hyperplastic mucosal stroma [12,25,26].
The management of rhinosporidiosis primarily includes surgical excision of the lesion combined with cauterization of the base to minimize recurrence which is significant clinically [18,27,28]. Adjuvant medical therapies like administration of dapsone, have been used with variable success, however complications such as autoimmune hemolytic anemia have been reported, necessitating cautious use [19]. Disseminated and recurrent rhinosporidiosis cases have therapeutic challenges which require customized approaches [18,19,28]. Novel surgical modalities and reconstructive techniques have been proposed to improve outcomes in difficult anatomical sites [29].
Orofacial rhinosporidiosis has been minimally discussed in literature with fragmented evidence scattered across few isolated case reports or series. The lack of a consolidated overview restricts the clinical understanding and obstructs the development of evidence-based strategies. This is due to the differences in presentation, progression, and prognosis of the disease from its nasal variant. This systematic review addresses these gaps by synthesizing published literature to establish a structured framework for diagnosis and management. The objectives are to clarify pathogenetic mechanisms contributing to orofacial disease, describe the range of clinical manifestations including uncommon presentations, assess the utility and accuracy of available diagnostic tools, and evaluate the role of surgical and pharmacological interventions. By integrating these domains, the review aims to support clinicians in making informed therapeutic decisions, minimize recurrence, and propose standardized care pathways that can serve as a reference for future research and clinical practice.
Methodology
Research Question and PICO/PICOS Framework: This systematic review has been registered at International Prospective Register of Systematic Reviews (PROSPERO) with PROSPERO ID CRD420251107401. It was guided by the research question: “What are the pathogenetic mechanisms, clinical features, and effective management strategies for orofacial rhinosporidiosis?” The review employed the PICO framework to structure the question as follows: Population (P): patients diagnosed with orofacial rhinosporidiosis; Intervention (I): various management approaches including surgical and medical treatments; Comparison (C): different treatment modalities or no treatment; and Outcomes (O): clinical resolution, recurrence rates, and complication incidence. Study design (S), incorporating case reports and case series to ensure comprehensive coverage of available evidence.
Search Strategy: The systematic search was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines to ensure transparency and reproducibility. Electronic databases were searched comprehensively, including PubMed, Scopus, Web of Science. Duplicates or irrelevant results were manually screened and removed, with all searches managed according to PRISMA 2020 recommendations. The timeline for literature retrieval spanned from 2012 to 2025. Key search terms included “rhinosporidiosis”, “orofacial rhinosporidiosis”, “oral cavity rhinosporidiosis”, “maxillary sinus rhinosporidiosis”, “buccal mucosa rhinosporidiosis”, “palatal rhinosporidiosis”, “nasopharyngeal rhinosporidiosis”, “oropharyngeal rhinosporidiosis”, “gingival rhinosporidiosis”, “tongue rhinosporidiosis”, “facial swelling rhinosporidiosis”, “management”, “treatment”, “surgical excision rhinosporidiosis”, “dapsone therapy rhinosporidiosis”, “recurrence rhinosporidiosis”, “disseminated rhinosporidiosis”, “diagnosis”, “histopathology rhinosporidiosis”, “clinical features”, and “pathogenesis”. Boolean operators such as AND, OR, and NOT were used to combine keywords strategically; for example, (“rhinosporidiosis” OR “orofacial rhinosporidiosis”) AND (“management” OR “treatment”).
Eligibility Criteria: Inclusion criteria comprised peer-reviewed articles, case reports and case series, focusing on orofacial rhinosporidiosis with clearly documented clinical, diagnostic, or therapeutic information. Case reports and series were included because orofacial rhinosporidiosis is uncommon, and these papers are most commonly available as clinical sources. These reports help to clarify diagnostic features and guide the management where large comparative studies are unavailable. Articles published in English were considered to ensure accurate interpretation. Exclusion criteria involved studies that focused exclusively on nasal or nasopharyngeal rhinosporidiosis without orofacial involvement, review articles without original data, conference abstracts lacking full-text availability, and non-human studies.
Selection Criteria: The initial screening of titles and abstracts was conducted independently by two reviewers (KC and AD) to exclude irrelevant studies. Full texts of potentially eligible articles were then retrieved and assessed for inclusion by the same reviewers. Any discrepancies were resolved through discussion or, if needed, adjudicated by a third reviewer (CS) to ensure impartial selection and consensus. A PRISMA flow diagram was applied to document the number of records identified, screened, included, and excluded at each stage, along with reasons for exclusion.
Data Extraction, Synthesis, and Quality Assessment: Out of a total of 238 articles, only 16 were included in the review after proper screening of the data (Fig. 1). Data extraction was performed independently by reviewers using a pre-designed standardized form capturing study characteristics, patient demographics, clinical presentation, diagnostic methods, treatment modalities, outcomes, and follow-up details. Case reports and case series were appraised using the Joanna Briggs Institute (JBI) critical appraisal checklist [30].
Risk of Bias and Quality Assessment: Risk of bias was assessed using the Joanna Briggs Institute (JBI) checklist, with scores interpreted as low, moderate, or high; studies with higher bias contributed only to narrative synthesis without affecting the main interpretations. Specific attention was paid to selection bias, reporting bias, and outcome ascertainment owing to the predominance of case reports and series in this rare condition. Studies with high risk of bias or methodological limitations were identified and their influence on the overall synthesis was considered [31]. The use of standardized appraisal tools facilitated objective evaluation.
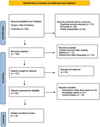 |
Fig. 1 PRISMA flowchart for the review. |
Results
Table I shows that most patients with orofacial rhinosporidiosis were young to middle-aged men, with only two female cases reported [18,27]. The age of affected individuals ranged from 10 to 65 yr [4,10,29,32–36]. Male predominance is commonly linked to environmental or occupational exposure, such as swimming or bathing in stagnant water, while female cases were less frequent [6,10,18,27,32,34]. The nasal cavity, maxillary sinus, and nasopharynx were the most frequently involved sites [4,6,27,29,32], whereas the oral cavity including the buccal mucosa, palate, and oropharynx was affected less often [2,3,35,36]. Rare disseminated cases involved subcutaneous tissues or lungs [33]. Clinically, lesions were typically polypoid, reddish, friable, and “strawberry-like” in appearance [2,3,6,27,35,36], with symptom duration ranging from a few weeks to 20 yr [4,29,32,36]. Long-standing lesions sometimes caused facial deformity, proptosis, or recurrent epistaxis [10,18,34], highlighting the need for careful diagnosis in endemic regions [23,31,34].
Table II shows that histopathology was consistently used to confirm orofacial rhinosporidiosis, demonstrating sporangia with endospores in all reported cases. Imaging modalities, such as CT scans, X-rays, and nasal endoscopy, were variably employed to delineate lesion extent [4,6,13,19,29,33,36], while some cases relied solely on clinical assessment [2,10]. Surgical excision with electrocauterization or endoscopic techniques was the primary intervention [3,6,13,27,34,35], often supplemented with Dapsone for recurrence prevention or as adjunctive therapy [4,13,27,29,36]. Recurrences were uncommon but occurred in select cases, particularly with disseminated disease or incomplete excision [4,29,32,33]. Follow-up durations ranged from 3 to 36 months [2,3,27,33], and complications were minimal, with rare instances of Dapsone-induced hemolytic anemia [33]. Overall, combined surgical and medical management achieved favorable outcomes [3,4,27,35].
Table III presents the methodological quality assessment of 16 case reports using a standardized tool for evaluating case reports and series [31]. Scores were based on selection, ascertainment (A1, A2), causality (C1–C4), and reporting criteria. Four studies achieved a high-quality rating [3,4,33,35], scoring 7/8 due to comprehensive patient selection, detailed diagnostic confirmation, and thorough follow-up. Most remaining reports were rated moderate-high [2,6,10,13,27,29,34,36], demonstrating adequate methodology but with occasional gaps such as incomplete causality assessment or missing follow-up details. Five studies were rated moderate [4,10,18,19,32], mainly due to partial reporting or limited selection criteria. This analysis highlights variability in case report quality, emphasizing the need for standardized reporting to improve reproducibility, diagnostic clarity, and outcome assessment in orofacial rhinosporidiosis.
Table IV presents the risk of bias assessment of individual case reports on orofacial rhinosporidiosis, evaluated using the Joanna Briggs Institute (JBI) Critical Appraisal Checklist for Case Reports. The tool evaluates methodological rigor across ten items, covering patient selection, diagnostic methods, intervention details, outcomes, and follow-up. Across the included studies, most reports scored “ + +” (low risk of bias) in Items 1–3 (patient demographics, clinical history, and diagnostic methods), indicating well-documented case descriptions. Item 4 (clear reporting of diagnostic tests) varied, with some studies marked as “NA” where imaging was not performed [3,29,36]. Item 5 (intervention clearly described) showed occasional gaps, particularly in older studies [29,34]. For Item 6 (adverse events reporting), several reports failed to provide details, receiving “–” [4,6,13,27,36]. Item 7 (takeaway message/conclusion) was consistently well-addressed, while Item 8 (follow-up adequacy) varied, with some studies limited by short-term follow-up [1,5,7]. Items 9 and 10 (outcomes measured and reported) were generally well-documented, showing consistent strengths across most reports. The majority of studies demonstrated moderate-to-high quality, with comprehensive patient details, diagnostic confirmation via histopathology, and clear outcome reporting. Methodological limitations were mainly linked to incomplete reporting of adverse events and variable follow-up duration. This suggests that while evidence from case reports remains valuable, future reports should strengthen consistency in documenting interventions, complications, and long-term outcomes to minimize bias.
Demographic and clinical characteristics of reported case reports of orofacial rhinosporidiosis.
Diagnostic modalities, therapeutic interventions, and outcomes in reported cases of orofacial rhinosporidiosis.
Risk of bias assessment individual studies - JBI's tool for case series.
Discussion
Rhinosporidiosis is a chronic granulomatous disease caused by Rhinosporidium seeberi, characterized by the formation of vascular, friable polyps in mucocutaneous regions, predominantly in the nose and oropharynx. The rare infection continues to pose diagnostic and therapeutic challenges due to its relapsing course and its ability to mimic other pathologies. The present review synthesized evidence from published case reports of orofacial rhinosporidiosis, with particular focus on clinical presentation, diagnostic approaches, management strategies, and recurrence patterns. Case reports and series were included because orofacial rhinosporidiosis is rare disease and most cases present the findings in an elaborate manner. These publications are the main clinical source for uncommon disease presentations and give practical information when there is deficient data from other sources [31].
The analysis of published case reports revealed that most patients with orofacial rhinosporidiosis were young to middle-aged men (with two cases reported in females) (Tab. I). This demographic pattern has been consistently observed in larger series from endemic regions, where male predominance was more than females with a ratio ≥ 2:1 [1,6,14,21]. The predominance of male is commonly attributed to environmental and occupational exposure, since swimming or bathing in stagnant pond water is a recognized risk factor for acquiring the infection. This review found fewer female cases, supporting earlier reports that the disease is less common in women but not limited to men [14]. The age distribution, which ranged from the 2nd to the 6th decade, also matches the trend reported in institutional data where most cases occur in the younger age groups, possibly revealing greater outdoor exposure during these years [8,13].
The commonest sites of involvement are the nasal cavity, paranasal sinuses, and nasopharynx which is consistent with the well-established predilection of R. seeberi for the upper respiratory tract mucosa, where trauma and microabrasions facilitate implantation of endospores [19,37]. Oral cavity involvement, though less frequent, was documented in some cases, confirming previous reports that oral mucosa can be directly infected or secondarily involved from contiguous spread [10,34]. Disseminated cases involving the lungs, skin, or subcutaneous tissue represent the extreme clinical spectrum and are rarely reported, but they highlight the organism's potential for hematogenous spread in advanced or recurrent disease [33,37]. Clinically, the lesions described in this review were characteristically polypoid, reddish, and friable, often likened to a strawberry in appearance. This vascular, granulation-like morphology is pathognomonic and has been consistently described across decades of literature [3,6,27,34]. The duration of symptoms varied widely, from weeks to several years, reflecting the slow-growing yet persistent nature of the infection. Long-standing lesions were often associated with facial deformity, proptosis, or recurrent epistaxis, depending on the site and size of the mass [2,8,32]. These findings emphasize the importance of considering rhinosporidiosis in the differential diagnosis of chronic nasal and oropharyngeal masses, especially in endemic settings, since the disease can mimic benign polyps, papillomas, or even malignancies [4,6,37].
The gold standard for diagnosis in all reviewed cases was histopathology, which demonstrated sporangia of varying sizes containing numerous endospores. This feature is highly distinctive and remains the only reliable diagnostic method, as attempts to culture R. seeberi have been unsuccessful [38–41]. In clinical practice, the inability to culture the organism emphasizes the critical role of biopsy and histopathological examination. The constant dependence on histology in both case reports and institutional studies reinforces its indispensability. The use of imaging modalities such as CT or MRI was different across the reviewed cases. Some reports included detailed radiological assessments to delineate lesion extent, particularly in sinonasal and orbital disease [3,6,29,42], whereas others relied solely on endoscopic or gross examination findings. The varying use of imaging reflects differences in reporting standards and resource availability, but it also creates inconsistency in how completely the disease was evaluated. In the quality assessment, reports that included imaging and complete clinical description scored higher for diagnostic clarity compared to those that relied exclusively on histopathology without detailing preoperative evaluation [26,43]. Another important aspect identified was the incomplete reporting of follow-up. While some reports documented outcomes for more than a year, others mentioned only short-term recovery. This variation restricts the dependability of recurrence data, since rhinosporidiosis is known for late recurrences (sometimes several years after surgery) [4,29]. Similarly, adverse event reporting was found to be often absent, with few reports addressing complications related to surgery or Dapsone therapy. This represents a significant methodological gap, because Dapsone, while useful in reducing recurrence, carries risks such as hemolytic anemia and methemoglobinemia [33,44]. Adverse event reporting was often missing, which reduced the value of these case reports. The methodological quality ranged from moderate to high. Reports with complete demographic data, diagnostic methods, surgical details, and long-term outcomes scored higher on JBI. Those with incomplete follow-up or poor safety reporting scored lower. Consistent use of standardized guidelines, such as the CARE checklist, can improve the reliability and comparability of published cases [30].
The findings of this review confirmed that surgical excision, often combined with electrocauterization of the base, remains the cornerstone of management for orofacial rhinosporidiosis. This is supported by previous reports, since meticulous removal of the lesion is essential to prevent recurrence [3,16,19,32]. However, surgical excision alone is not always curative, especially in cases where complete clearance is difficult due to anatomical constraints or lesion size. Adjunctive therapy with Dapsone was widely reported in the reviewed cases, with most authors justifying its use based on the drug's ability to inhibit maturation of sporangia and promote fibrosis of lesion stroma [8,10,21,34]. In many cases, patients who received post-operative Dapsone remained recurrence-free during follow-up. However, recurrences still occurred in some cases despite medical therapy, which may reflect incomplete surgical removal or variability in individual drug responsiveness [4,20,29]. This highlights that Dapsone is not a substitute for surgery but serves as a valuable adjunct. The exceptional cases of disseminated disease revealed an even greater therapeutic challenge. In such patients, conventional surgery is impractical, and pharmacological approaches have been explored. Some reports described attempts with antifungals or broad-spectrum antibiotics, but outcomes were inconsistent, and no drug has proven reliably effective in systemic disease [15,33,38]. These rare but severe cases emphasize the urgent need for research into alternative therapeutic strategies beyond the current surgical-medical model. An important observation in this review was the inadequacy of long-term follow-up in many reports. Since recurrence is a hallmark of rhinosporidiosis, often occurring months or years after treatment, short-term follow-up cannot be equated with cure. Studies that documented outcomes for more than a year provided more meaningful insights into recurrence rates, while those with only brief follow-up risked underestimating the true burden of relapse [17,41]. This highlights the significance of structured, long-term monitoring in affected patients, with follow-up extending beyond the immediate postoperative period.
The synthesis of case reports in this review reaffirms the established principles of rhinosporidiosis management but also exposes persistent gaps in evidence quality. The dependence on histopathology for diagnosis remains unchallenged, and surgery with adjunctive Dapsone continues to represent the most effective strategy for localized disease. However, recurrence persists as a challenge, partly due to incomplete excision, difficulty accessing deep sites, or inadequate follow-up. The review highlights the importance of suspecting rhinosporidiosis in patients presenting with chronic nasal or orofacial polyps, particularly in endemic regions. The classical strawberry-like morphology should prompt biopsy, and clinicians should aim for complete excision with electrocauterization, followed by medical therapy when appropriate. Structured follow-up extending beyond one year should be standard practice, given the risk of late recurrence. This review also highlights several unmet needs. Future case reports should adhere to standardized reporting frameworks to ensure completeness and reproducibility. Systematic documentation of adverse events, particularly with Dapsone, is necessary to balance efficacy with safety. Innovative therapeutic options for disseminated rhinosporidiosis warrant investigation, as conventional management remains inadequate for systemic disease.
Strengths and limitations: This review systematically synthesized evidence on orofacial rhinosporidiosis and provided the first structured framework on various clinical and management aspects. The strengths include comprehensive databases searched, strict eligibility criteria, and standardized quality assessment using the JBI checklist, which ensured methodological rigor. The review highlights consistent patterns of clinical features, confirms histopathology as the diagnostic gold standard, and evaluates combined surgical and pharmacological strategies. However, limitations arise from dependence on only case reports and small case series, which are prone to reporting bias, incomplete follow-up, and underreporting of adverse events. The geographic concentration of cases in endemic regions also restricts the generalizability. Future multicenter research is crucial to refine management strategies and improve outcomes in this neglected infection. Future studies should define clear diagnostic criteria, test treatment methods across centres, and set up regional records to monitor cases and patient recovery.
Conclusion
Hence, the present review reports published evidence on orofacial rhinosporidiosis and shows its relevance in routine clinical practice. Clinicians should consider the disease when patients present with strawberry-like polyps in the oral or sinonasal region and confirm diagnosis through histopathology, as imaging alone often proves inconclusive. Surgical excision with cauterization remains the mainstay of management, and adjunctive Dapsone may reduce recurrence, though safety monitoring is essential. The review highlights certain challenges, including late recurrences, inadequate reporting of adverse events, and the absence of effective options for disseminated disease. Standardized reporting using the CARE checklist, structured long-term follow-up, and exploration of novel pharmacological or immunological therapies are strongly recommended.
Funding
This study was self-funded.
Conflicts of interest
The authors declare that they have no conflict of interest.
Data availability statement
Not applicable as the topic is a systematic review and all the data used is available on the internet.
References
- Rath R, Baig SA, Debata T. Rhinosporidiosis presenting as an oropharyngeal mass: a clinical predicament? J Nat Sci Biol Med 2015;6:241–245. [Google Scholar]
- Santosh BS, Kumar AH, Singh R, Jose J, Shivamrthy DM, Daniel D. A rare presentation of rhinosporidiosis on buccal mucosa− a case report. J Oral Med Oral Surg 2022;28:5. [Google Scholar]
- Swain SK. Rhinosporidiosis presenting as a large oral cavity mass: a case report. Apollo Med 2025;22:S16–S19. [Google Scholar]
- Tong TK, Ismail I, Yunus Mohammad NM, Yusoff SM, Sahri AM. Recurrent rhinosporidiosis: a case report from Malaysia and review of literature. Proc Singapore Healthcare 2023 Feb 24;32:20101058231160606. [Google Scholar]
- Penagos S, Zapata N, Castro JJ, Hidron A, Agudelo CA. Rhinosporidiosis in the Americas: a systematic review of native cases. Am J Trop Med Hyg 2021 17;105:171–175. [Google Scholar]
- Arias AF, Romero SD, Garcés CG. Case report: rhinosporidiosis literature review. Am J Trop Med Hyg 2020 7;104:708–711. [Google Scholar]
- Bezemer JM, Meesters K, Naveda CL, Machado PRL, Calvopiña M, Leeflang MMG, Schallig HDFH, de Vries HJC. Clinical criteria for Mucosal Leishmaniasis diagnosis in rural South America: a systematic literature review. PLoS Negl Trop Dis 2022 10;16:e0010621. [Google Scholar]
- Bhargava S, Grover M, Maheshwari V. Rhinosporidiosis: intraoperative cytological diagnosis in an unsuspected lesion. Case Rep Pathol 2012;2012:101832. [Google Scholar]
- Saha J, Basu AJ, Sen I, Sinha R, Bhandari AK, Mondal S. Atypical presentations of rhinosporidiosis: a clinical dilemma? Indian J Otolaryngol Head Neck Surg 2011 Jul;63:243–246. [Google Scholar]
- Putthia H, Manjunatha BS, Astekar M, Taufiq S. Palatal rhinosporidiosis: an unusual case report and review of the literature. J Korean Assoc Oral Maxillofac Surg 2018 ;44:293–297. [CrossRef] [PubMed] [Google Scholar]
- Gupta RK, Singh BP, Singh BR. Rhinosporidiosis in Central India: a cross-sectional study from a tertiary care hospital in Chhattisgarh. Trop Parasitol 2020 Jul-;10:120–123. [Google Scholar]
- Mahmud S, Haque R, Al Mamun A, Alam R, Akaiduzzaman DG, Datta UK, Alam Z. A clinicopathological study of Rhinosporidiosis. Bangladesh J Otorhinolaryngol 2015;21:94–96. [Google Scholar]
- Kazi AZ, Kazi HZ, Channe RC, Kulkarni AS. Rhinosporidiosis of maxillary sinus: a case report. J Indian Acad Oral Med Radiol 2019 Jul 1;31:274–279. [Google Scholar]
- Banjara H, Satyapal K, Ravi AK, Kaur J, Rohilla S, Dhaulakhandi DB. Rhinosporidiosis: a report of 104 cases from Raipur, Chhattisgarh (India). Trop Parasitol 2025;15:12–20. [Google Scholar]
- Salim T, Komu F. Varied presentations of cutaneous rhinosporidiosis: a report of three cases. Indian J Dermatol 2016-Apr;61:209–212. [Google Scholar]
- Singh I, Singh A, Gupta V, Goyal S, Kumar M. Recurrent nasal and disseminated rhinosporidiosis. Glob J Otolaryngol 2017;6:555691. [Google Scholar]
- Behera S, Chowdhury RK, Dora J. Rhinosporidiosis of the lacrimal sac in a tertiary care hospital of India-A retrospective case study. Indian J Ophthalmol 2022 1;70:1732–1735. [Google Scholar]
- Rameshkumar A, Gnanaselvi UP, Dineshkumar T, Raghuram PH, Bharanidharan R, Rajkumar K. Rhinosporidiosis presenting as a facial swelling: a case report. J Int Oral Health 2015 Feb;7:58–60. [Google Scholar]
- Dey AK, Sharma R, Mittal K, Kumar P, Murumkar V, Mitkar S, Hira P. Rhinosporidiosis: a rare cause of proptosis and an imaging dilemma for sinonasal masses. Case Rep Otolaryngol 2016;2016:3573512. [Google Scholar]
- Chen L, Buonocore D, Wang B, Tabaee A. Delayed recurrence of sinonasal rhinosporidiosis. Am J Otolaryngol 2015 Nov 1;36:778–780. [Google Scholar]
- Bhat V. Comments on 'novel multidrug therapy for disseminated rhinosporidiosis, refractory to dapsone - case report'. Trop Doct 2014 Jan;44:59–60. [Google Scholar]
- Raja K, Thangavel S, Kushwaha A, Srinivas BH, Kar R, Alexander A, Penubarthi LK, Saxena SK. Management of disseminated rhinosporidiosis: experience from a single tertiary institution. Turk Arch Otorhinolaryngol 2024 Oct 23;62:66. [Google Scholar]
- Bandopadhyay SN, Jana U, Bandopadhyay G, Majhi TK, Sen S, Das S, Mandal I. Rhinosporidiosis: various presentations and different sites. Bengal J Otolaryngol Head Neck Surg 2015 1;23:48–56. [Google Scholar]
- Aroor R, Gowda MM, Bhat VS, Bhandary SK. Novel approach to rhinosporidiosis. Int J Otorhinolaryngol Clin 2014 1;6:55–57. [Google Scholar]
- Mathew S, Arora RD, Prabha N, Kamble P, Satpute SS, Nagarkar NM. Retroanalytical study of epidemiological factors of rhinosporidiosis. Int Arch Otorhinolaryngol 2020 Nov 30;25:e504–e508. [Google Scholar]
- Hossain T, Hanif MA. Histopathological spectrum of sinonasal masses in patients coming to a tertiary care hospital: a study of 150 cases. Saudi J Med Pharm Sci 2023;9:56–60. [Google Scholar]
- Doddawad VG, Singh R, S S. A new technique to resolve Nasal Rhinosporidiosis: a case report with review of literature. Int J Surg Case Rep 2022;92:106807. [Google Scholar]
- Majumdar AB, Biswas D, Paul SS, Ray S, Kumar G. Rhinosporidiosis: a clinicopathological study from a Rural Tertiary Health Care Centre, Bihar. Int J Adv Med 2014;1:213–216. [Google Scholar]
- Ali GM, Goravey W, Al Hyassat SA, Petkar M, Al Maslamani MA, Hadi HA. Recurrent nasopharyngeal rhinosporidiosis: case report from Qatar and review of the literature. IDCases 2020 Jul 3;21:e00901. [Google Scholar]
- Munn Z, Barker TH, Moola S, Tufanaru C, Stern C, McArthur A, Stephenson M, Aromataris E. Methodological quality of case series studies: an introduction to the JBI critical appraisal tool. JBI Evid Synth 2020 Oct;18:2127–2133. [Google Scholar]
- Murad MH, Sultan S, Haffar S, Bazerbachi F. Methodological quality and synthesis of case series and case reports. BMJ Evid Based Med 2018;23:60–63. [Google Scholar]
- Babu S, Anuradha A, Chandra S, Kashyap B. Rhinosporidiosis: a case report with review of literature. Ann Trop Med Public Health 2012 1;5. [Google Scholar]
- Ghosh R, Mondal S, Roy D, Ray A, Mandal A, Benito-León J. A case of primary disseminated rhinosporidiosis and dapsone-induced autoimmune hemolytic anemia: a therapeutic misadventure. IDCases 2021 Jan 1;24:e01076. [Google Scholar]
- Shah R. Rhinosporidiosis of maxillary sinus: an unusual site–a case report. Asian Arch Pathol 3: 5–12. [Google Scholar]
- Chandran A, Sakthivel P, Kumar R, Rajeshwari M. Pharyngeal rhinosporidiosis. BMJ Case Rep 2020 17;13:e236404. [Google Scholar]
- Devaraja K, Sagar P, Singh CA, Kumar R. Nondisseminated rhinosporidiosis with multisite involvement in the head and neck. Ear, Nose Throat J 2018 Sep;97:E15–E17. [Google Scholar]
- Izimukwiye AI, Mbarushimana D, Ndayisaba MC, Bigirimana V, Rugwizangoga B, Laga AC. Cluster of Nasal Rhinosporidiosis, Eastern Province, Rwanda. Emerg Infect Dis 2019 Sep;25:1727–1729. [Google Scholar]
- Idirisinghe KA, Sumanasena JA, Madarasinghe N. Disseminated rhinosporidiosis following spontaneous regression of the possible primary lesion. J Diagn Pathol 2016 Oct 19;11. [Google Scholar]
- Dutta S, Haldar D, Dutta M, Barik S, Das Biswas K, Sinha R. Socio-demographic correlates of rhinosporidiosis: a hospital-based epidemiologic study in Purulia, India. Indian J Otolaryngol Head Neck Surg 2017;69:108–112. [Google Scholar]
- Almeida FA, Feitoza Lde M, Pinho JD, Mello GC, Lages JS, Silva FF, Silva RR, Silva GE. Rhinosporidiosis: the largest case series in Brazil. Rev Soc Bras Med Trop 2016 Jul-Aug;49:473–476. [Google Scholar]
- Almeida FA, Teixeira-Junior AAL, Pinho JD, Costa EF, Silva GEB. Evaluation of diagnosed cases of eye rhinosporidiosis in a public hospital ofanhão, Northeast Brazil. BMC Ophthalmol 2019 Nov 8;19:218. [Google Scholar]
- John D, Selvin SST, Irodi A, Jacob P. Disseminated rhinosporidiosis with conjunctival involvement in an immunocompromised patient. Middle East Afr J Ophthalmol 2017 Jan-Mar;24:51–53. [PubMed] [Google Scholar]
- Maheshwari A, Bansal A. Clinico-pathological spectrum of sinonasal masses: a tertiary care hospital experience. Int J Otorhinolaryngol Head Neck Surg 2017 Oct;3:1015–1019. [Google Scholar]
- Singh R, Nand A. An epidemiological study of rhinosporidiosis in a tertiary care center in Rajnandgaon, Chhattisgarh. Int J Otorhinolaryngol Head Neck Surg 2021 Jan;7:95–100. [Google Scholar]
Cite this article as: Chandrasekaran K, Sridhar C, Deshpande A, 2026. Etiology, clinical features, and management strategies in orofacial rhinosporidiosis: a systematic review. J Oral Med Oral Surg. 32: 7. https://doi.org/10.1051/mbcb/2025042
All Tables
Demographic and clinical characteristics of reported case reports of orofacial rhinosporidiosis.
Diagnostic modalities, therapeutic interventions, and outcomes in reported cases of orofacial rhinosporidiosis.
All Figures
 |
Fig. 1 PRISMA flowchart for the review. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


