| Issue |
J Oral Med Oral Surg
Volume 32, Number 1, 2026
|
|
|---|---|---|
| Article Number | 4 | |
| Number of page(s) | 7 | |
| DOI | https://doi.org/10.1051/mbcb/2026006 | |
| Published online | 20 March 2026 | |
Original Research Article
Evaluation of pedicled buccal pad of fat with zygomatic implants in reducing vestibular gingival recession: a pilot study
1
Surgery Department, Faculty of Dentistry, Cairo University, Egypt
2
Oral and Maxillofacial Surgery Department, Faculty of Dentistry, Cairo University, Egypt
* Correspondence: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
8
September
2025
Accepted:
29
January
2026
Abstract
Background: The rehabilitation of severely atrophic maxillae with extra-maxillary zygomatic implants often poses challenges due to insufficient soft tissue volume, resulting in implant body exposure and increased risk of gingival recession, peri-implant mucositis, and chronic inflammation, which can potentially compromise implant survival. Objective: This prospective clinical split-mouth study aimed to evaluate the effectiveness of the buccal fat pad flap (BFPF) in minimizing gingival recession and improving peri-implant soft tissue outcomes in patients receiving extra-maxillary zygomatic placement for severe maxillary atrophy. Methods: Four patients presenting with Cawood and Howell Class IV–VI maxillary atrophy were rehabilitated using the quad-zygomatic implant technique. Each patient received bilateral zygomatic implants; on one side, the implant was covered with a buccal fat pad (BFP) over the externalized implant, while the contralateral side served as a control and underwent conventional mucoperiosteal soft-tissue closure. Gingival recession, peri-implant tissue conditions, modified bleeding index (mBI), presence of suppuration, and implant survival were assessed at 3, 6, and 12 months postoperatively. Results: The BFPF group demonstrated significantly less gingival recession at all follow-up intervals (P < 0.05) and lower mBI scores. Suppuration was absent in both groups. Although harvesting time for the BFPF was slightly longer, it did not significantly prolong the overall surgery. Conclusion: The pedicled buccal fat pad flap is a promising adjunct for enhancing soft tissue outcomes in atrophic maxillae rehabilitation, reducing gingival recession, and reducing peri-implant soft tissue complications.
Key words: Zygomatic implants / extra-maxillary approach / buccal fat pad flap / gingival recession / peri-implant soft tissue / maxillary atrophy
© EDP Sciences, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Extra-maxillary zygomatic implants (EZIs) are a surgical protocol in which implants are anchored in the zygomatic bone through the alveolar ridge or buccal side, bypassing the maxillary sinus. This approach provides a less invasive alternative to traditional intra-sinus techniques [1]. This approach reduces sinus-related complications, such as chronic sinusitis. However, it increases the risk of peri-implant soft tissue recession, dehiscence, implant exposure, and peri-implantitis [2,3]. According to the zygomatic anatomy-guided approach (ZAGA) classification, ZAGA Types 3–4 patients, who have significant maxillary bone loss and concave lateral maxillary walls, are ideal candidates for extra-maxillary zygomatic implant placement because their anatomical configuration renders an intra-sinus zygomatic implant approach less feasible [4,5].
The buccal fat pad (BFP) is a specialized, encapsulated adipose structure commonly used for intraoral reconstruction following tumor resection [6,7]. It offers numerous advantages, being easily harvestable, well-vascularized, associated with minimal donor site issues, and capable of forming tissue like normal oral mucosa within a few weeks [8], and it is associated with a lower risk of postoperative complications, such as infection or hematoma [9].
These advantages make the BFP a valuable option to mitigate soft tissue breakdown, reduce the risk of peri-implant complications, and improve both functional and esthetic outcomes in patients with severe maxillary atrophy [10–12]. However, the effectiveness of applying the BFP as an adjunct in zygomatic implant–supported prosthetic rehabilitation to overcome complications associated with the extra-maxillary technique, particularly soft tissue recession, remains unclear. This is a split-mouth study, and to the best of our knowledge, no previous split-mouth investigation has evaluated whether the use of the buccal fat pad for coverage of extra-maxillary implants yields superior outcomes compared with conventional soft-tissue closure techniques.
Therefore, the present study aimed to investigate whether using a pedicled fat pad with EZIs could effectively prevent or reduce gingival recession compared with standard extra-maxillary placement.
Materials and methods
Study design/sample
This prospective, split-mouth clinical study evaluated 16 zygomatic implants placed in four patients presenting with Cawood and Howell Class IV–VI maxillary atrophy. The side designated for the test intervention was randomized preoperatively, with the contralateral side serving as the control. All procedures were performed at the Department of Oral and Maxillofacial Surgery, Faculty of Dentistry, Cairo University, Egypt, between August 2024 and April 2025. Written informed consent was obtained from all participants. The study protocol was approved by the Cairo University Ethics Committee in 2023 (approval number: 5-7-23).
Patients over 18 years of age, of both sexes, with Cawood and Howell Class IV–VI maxillary atrophy were included in this study. Participants needed to be in good systemic health and committed to complying with scheduled follow-up visits. Exclusion criteria comprised individuals with systemic conditions that could impair hard or soft tissue healing, such as uncontrolled diabetes mellitus, or those taking medications known to negatively affect wound repair, including long-term corticosteroids or immunosuppressive agents. Patients with intrabony lesions or active oral infections, detrimental habits such as smoking, and a history of head and neck radiotherapy within the past 10 years were also excluded.
The 16 zygomatic implants were divided into two groups: the control group, which included 8 implants placed using the standard extra-maxillary zygomatic implant technique, and the intervention group, which included 8 implants placed using a pedicled pad of fat in conjunction with EZIs.
Surgical procedure
All surgical procedures were performed by the same operator. Postoperative clinical evaluations were conducted by a different oral and maxillofacial surgeon from the same department, and this evaluator remained consistent throughout the study to minimize assessment bias.
All patients underwent a standardized preoperative protocol that included detailed history taking, clinical examination, fabrication of diagnostic acrylic dentures, and acquisition of a preoperative cone-beam computed tomography (CBCT) scan. CBCT was performed using the Planmeca ProMax® 3D Mid unit (Planmeca Oy, Helsinki, Finland) at a voxel size of 0.3 mm. Digital imaging data were imported into Blue Sky Plan® software 4.0 (Blue Sky Bio, LLC, USA) for virtual surgical planning [13] (Fig. 1), with bilateral zygomatic implants placed using the quad zygomatic technique in fully edentulous cases.
All patients underwent surgery under general anesthesia with nasal endotracheal intubation. Local anesthesia consisting of 2% lidocaine with 1:100,000 adrenaline (Arab Drug Company for Pharmaceuticals, Cairo, Egypt) was infiltrated intraorally along the planned incision lines to achieve hemostasis. Surgical procedures involved the elevation of a full-thickness mucoperiosteal flap, exposure of the zygomatic buttress, and placement of JD Zygoma implants (JDentalCare Srl, Modena, Italy) with diameters of 3.9 and 4.3 mm and lengths ranging from 30 to 57.5 mm. The JD implant is designed with a smooth cervical body and a threaded, tapered apex. The implants were inserted following an extra-maxillary trajectory that was virtually simulated using Blue Sky software, guided by anatomical landmarks as described by Rigolizzo et al. [14] and Rossi et al. [15] (Fig. 2). Implants were fitted with abutment screws and healing abutments.
In the intervention group, a pedicled buccal fat pad was harvested and advanced to cover the exposed implant surface, providing vascularized soft tissue protection. The flap was sutured to the palatal mucosa (Fig. 3). Flaps were sutured using resorbable sutures to ensure a tension-free closure. Vicryl® sutures 4-0 (polyglactin 910) on a non-cutting round needle (Ethicon, Johnson & Johnson, USA) were used to secure the pedicled buccal fat pad flap, while chromic catgut 4-0 (Ethicon, Johnson & Johnson, USA) was used for mucosal suturing. Postoperative care included amoxicillin/clavulanic acid 625 mg every 8 h for 5 days (Augmentin®, GlaxoSmithKline, UK), ibuprofen 600 mg every 8 h for 3 days (Brufen®, Abbott Laboratories, USA) for pain control, and 0.12% chlorhexidine gluconate mouthwash twice daily for 14 days (Corsodyl®, GlaxoSmithKline, UK).
Patients were followed up at 1 week, 3 months, 6 months, and 12 months. The zygomatic implants were left unloaded during the healing phase, with no provisional implant-supported prosthesis; some patients wore a pre-existing complete denture for temporary esthetic and functional replacement. At 6 months, prosthetic rehabilitation was initiated. A CBCT scan was obtained, and digital impressions were captured using scan bodies to digitally fabricate a full-arch implant-supported hybrid prosthesis. The primary outcome was gingival recession measured at 3, 6, and 12 months postoperatively using clinical parameters and 6-month CBCT, including peri-implant mucosal recession (PISTR), peri-implant soft tissue condition (PISTC), modified bleeding index (mBI), suppuration, and implant survival rate. The secondary outcome was the effect of harvesting on surgical time. The study variables were assessed using the criteria established by Hernaández-Alfaro et al. [11].
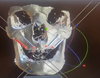 |
Fig. 1 Virtual preoperative planning for zygomatic implants using Blue Sky software. |
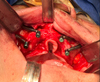 |
Fig. 2 Quad extra-maxillary zygomatic implants in situ, with healing abutments in place. |
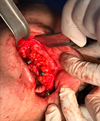 |
Fig. 3 The pedicled buccal fat pad flap covered the zygomatic implants on one side and was sutured to the palatal mucosa with resorbable sutures. |
Outcome assessment criteria
The implant survival rate was defined as the number of implants remaining stable without mobility. The PISTR was assessed through clinical examination and categorized as follows: Grade I = no recession, Grade II = slight recession with the implant head visible, and Grade III = recession with up to seven exposed threads. The PISTC was also evaluated clinically and scored as follows: 0 = normal mucosa, 1 = minimal inflammation with minor edema, 2 = moderate inflammation with edema and glazing, and 3 = severe inflammation with bleeding without probing. The mBI was determined by inserting a UNC-15 periodontal probe (Hu-Friedy) 1 mm into the sulcus surrounding the implant. Bleeding was evaluated 15 s after gentle probing and recorded on a scale from 0 to 3, where 0 = no visible bleeding, 1 = isolated bleeding spot, 2 = blood forming a confluent red line along the gingival margin, and 3 = heavy or profuse bleeding. Suppuration was assessed by applying gentle finger pressure to the peri-implant mucosa and was recorded as either present or absent.
Statistical analysis
Statistical analysis was performed using SPSS v26 (IBM Inc., Chicago, IL, USA). Shapiro-Wilk was used to evaluate the normality of data. Quantitative parametric data were presented as mean and standard deviation (SD). Qualitative variables were presented as frequency and percentage (%). Student’s t-test was used to analyze the differences between continuous data. The chi-square test (χ2) was used for comparison of categorical data. Statistical significance was set at P ≤ 0.05.
Results
This study included 16 zygomatic implants in 2 males and 2 females, with a median age of 55.9 yr. All patients were diagnosed with Cawood and Howell Class IV–VI maxillary atrophy and were classified as ZAGA III–IV. The 16 zygomatic implants were divided into two groups: control group: 8 implants placed using the standard extra-maxillary zygomatic implant technique, and intervention group: 8 implants placed using a pedicled pad of fat in conjunction with the externalized zygomatic.
The intergroup comparison between the standard EZI and the BFPF approach revealed comparable implant survival rates, with both groups achieving 100% survival and no statistically significant difference (P = 1.000). However, significant differences were observed in peri-implant soft tissue outcomes: the BFPF group demonstrated significantly less soft tissue recession across all time points. At 3 months (P = 0.005) (Fig. 4), at 6 months (P = 0.007), and at 12 months (P = 0.001) (Fig. 5). Similarly, PISTC was significantly better in the BFPF group at 6 months (P = 0.007) and at 12 months (P = 0.014) (Tab. I). Additionally, the BFPF group also exhibited significantly lower mBI scores than the EZI group at 3 months (P = 0.016), at 6 months (P = 0.005), and at 12 months (P = 0.005). Regarding suppuration, no suppuration was observed in either group at any follow-up time point; consequently, there were no statistically significant differences between groups (P > 0.05) (Tab. II).
The difference in operative time between the two surgical procedures was statistically significant (P < 0.0001). Although there was a minor increase in time, the mean surgery time for the BFPF technique was significantly longer than that of the standard EZI approach (59.87 vs. 65.25 min).
Comparison between the study groups regarding implant survival rate, PISTR, and PISTC
Comparison between the study groups regarding mBI and Suppuration
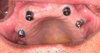 |
Fig. 4 Clinical photograph taken three months postoperatively illustrating the right side, where the BFPF was utilized to cover the EZI, demonstrating enhanced mucosal coverage and increased soft tissue thickness compared with the contralateral side (left), which exhibits visible gingival recession. |
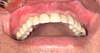 |
Fig. 5 A six-month postoperative intraoral photograph shows sustained mucosal coverage and increased soft tissue thickness on the right side treated with the BFPF, contrasting with gingival recession on the left side. |
Discussion
BFPF technique involves using the patient's buccal fat pad, located within the masticatory space between the buccinator and masseter muscles, to cover damaged areas in oral reconstruction and to fill larger maxillary defects [16,17]. The BFP volume tends to remain relatively constant throughout adulthood, ranging between approximately 7 and 11 cm³ in adults, while elderly people show similar or increased BFP volume [18,19]. However, anatomical and radiological studies have demonstrated that several factors, such as age, sex, body mass index, nutritional status, and medical history, can influence its size and quality [19].
BFP transposition is a single-use procedure; once the fat pad has been mobilized and sutured to the repair site, it cannot be re-harvested laterally due to flap necrosis, infection, or peri-implant complications. This underscores the importance of careful surgical planning when considering BFP transposition [18].
In the present study, the intergroup comparison between the EZI and BFPF techniques demonstrated that both achieved 100% implant survival with no significant differences, indicating equal mechanical stability. Regarding the implant survival rate, our results came following previous research that stated that both BFPF and EZI methods reported a 100% implant survival rate, indicating that the BFPF technique does not compromise the success of the implants, confirming that both techniques are mechanically reliable and capable of achieving successful osseointegration, even in severely atrophic maxillae [10,20].
Regarding soft tissue recession, the BFPF demonstrated significantly better outcomes in soft tissue recession across all time points, indicating improved soft tissue coverage and protection over time. This highlights the protective role of the BFPF in maintaining mucosal volume and stability. Current research agrees with our findings, indicating that using the BFPF to cover extra-maxillary zygomatic implants results in a statistically significant reduction in soft tissue recession over 12 months compared with EZI approaches [11]. In the same context, larger retrospective studies and systematic reviews supported these findings, reporting very low rates of tissue retraction and implant thread exposure when the BFPF technique is used, compared with higher risks of recession and exposure with EZI placement [21].
According to soft tissue conditions, the BFPF group maintained healthier peri-implant conditions at both 6- and 12-month follow-ups compared with the EZI group, suggesting that the buccal fat pad’s vascularity, biological activity, and cushioning properties may contribute to reduced inflammation and enhanced soft tissue integration. These results are in agreement with Chrcanovic & Abreu [22] and Aparicio et al. [23], who highlighted the challenges of soft tissue management in EZI and suggested that inadequate soft tissue coverage increases the risk of soft tissue infection, dehiscence, mucositis at the abutment level, and sinusitis. Additionally, Aparicio & Aparicio [24] and Leroy et al. [25] reported that the buccal fat pad provides stable, vascularized coverage that improves soft tissue healing and reduces postoperative complications when used in maxillofacial reconstruction and implantology. Furthermore, Abdel-Fatah et al. [21] and Elwan et al. [26] reported that BFPF increased the thickness of the peri-implant soft tissue and contributed to better long-term implant stability, which protects against mucosal dehiscence, inflammation, and long-term exposure of the implant surface to the oral cavity conditions that can lead to peri-implantitis.
In the present study, the BFPF group demonstrated significantly lower mBI scores at all evaluated time points compared with the EZI group, reflecting healthier peri-implant soft tissues and reduced inflammatory response. To the best of our knowledge, this is the first investigation to assess the influence of the BFPF on mBI scores in comparison with conventional EZI closure, and the findings suggest a promising role of the BFPF in reducing peri-implant bleeding and inflammation. The lower bleeding scores observed in the BFPF group may be attributed to the flap’s ability to enhance soft-tissue thickness, reduce soft-tissue recession and implant exposure, and promote more stable healing, ultimately lowering rates of complications such as bleeding [10,27].
In terms of surgery time, our results found that the BFPF technique required a statistically significantly longer surgical time compared with the EZI placement. However, this difference (approximately 5.4 min) is statistically significant, but it is still clinically acceptable. This difference is attributed to the complexity and additional steps, including harvesting and securing the pedicled BFP. Importantly, this slight increase in operative duration is compensated by the clinical benefits observed, including reduced soft tissue recession, lower bleeding scores, and improved peri-implant soft tissue health over the follow-up period. Our results are supported by a study that found the BFPF technique involves isolating and mobilizing the fat pad while preserving its vascular pedicle, which is crucial for flap viability and requires careful dissection and positioning, potentially extending surgical time [28].
Conclusion
The pedicled buccal fat pad offers a biologically effective adjunct to extra-maxillary zygomatic placement, significantly reducing gingival recession and inflammation while preserving peri-implant soft tissue integrity. Despite a modest increase in operative time, this approach enhances long-term clinical and esthetic outcomes in patients with severe maxillary atrophy.
Limitations
The small sample size of this pilot study limits the generalizability of the findings and the long-term assessment of outcomes. Therefore, larger-scale longitudinal studies are needed to validate these preliminary results and to better understand the variability in clinical outcomes.
Funding
The authors did not receive any funding for this article.
Conflicts of interest
The authors declare that they have no conflict of interest related to the products or information included in this article.
Data availability statement
The data that support the findings of this study are available from the corresponding author upon request.
Ethics approval
This study was reviewed and approved by the Research Ethics Committee. All procedures were conducted in accordance with the principles outlined in the Declaration of Helsinki.
Informed consent
Written informed consent was obtained from each participant to use the data and results for academic dissemination.
References
- Kämmerer PW, Heimes D, Hartmann A, Kesting M, Khoury F, Schiegnitz E, et al. Clinical insights into traumatic injury of the inferior alveolar and lingual nerves: a comprehensive approach from diagnosis to therapeutic interventions. Clin Oral Investig 2024;28:216. [Google Scholar]
- Tomaselli L. Quad zygomatic implants: case series. Ann Stomatol 2024;4:3. [Google Scholar]
- Nunes M, de Araújo NobreM, Camargo V. All-on-4 hybrid with extra-long transnasal implants: descriptions of the technique and short-term outcomes in three cases. J Clin Med 2024;13:3348. [Google Scholar]
- Aparicio C. The zygoma anatomy-guided approach: ZAGAa patient-specific therapy concept for the rehabilitation of the atrophic Maxilla. In: Zygomatic Implants, Cham: Springer International Publishing, 2020:63–85. [Google Scholar]
- Aparicio C, Olivo A, de Paz V, Kraus D, Luque MM, Crooke E, et al. The zygoma anatomy-guided approach (ZAGA) for rehabilitation of the atrophic maxilla. Clin Dent Rev 2022;6:2. [Google Scholar]
- Singh T, Kumar N, Sharma A. Evaluation of versatility and effectiveness of pedicled buccal fat pad for reconstruction of oral defects. Int J Health Sci (Qassim) 2021;5:334–342. [Google Scholar]
- Jolly SS, Singh A, Rattan V. The buccal fat pad as a primary flap for the reconstruction of intraoral defect after resection of oral cavity malignant tumors: a retrospective study. J Craniofac Surg 2024;35:e109–e111. [Google Scholar]
- Pizzolante T, Rasicci P, Saggiomo AP, Principi M, Capogreco M, Mummolo S. Buccal fat pad flap and buccal advancement flap for closure of oroantral fistula: a systematic review and a case report. Oral Implantol (Rome) 2024;16:50–61. [Google Scholar]
- Mallesh N, Akshatha MV, Hussien H. Study of the efficacy of pedicled buccal fat pad graft technique in the management of oro-antral communications. Int J Dent Res 2020;5:7–13. [Google Scholar]
- Blanco-Ruiz S, Molinero-Mourelle P, Blanco-Ruiz M, Fernández-Tresguerres F, Blanco-Samper S, López-Quiles J. Effect of the buccal fat pad in the prevention of zygomatic implant surgery postoperative complications: a pilot study. Med Oral Patol Oral Cir Bucal 2023;28:e371–e377. [Google Scholar]
- Alfaro F, Ragucci G, Valls-Ontańón A, Hamawandi A, Bertos-Quílez J. Extramaxillary zygomatic implant coverage with a pedicled buccal fat pad flap through a tunnel approach: a prospective case series. Int. J. Oral Maxillofac Implants 2022;37:400–406. [Google Scholar]
- Rahpeyma A, Khajehahmadi S. Buccal fat pad graft in maxillofacial surgery. Indian J Surg Oncol 2021;12:1–6. [Google Scholar]
- Collier J, Dawood A. The reverse zygomatic implant: a new implant for maxillofacial reconstruction. Br J Oral Maxillofac Surg 2016;54:e110. [Google Scholar]
- Rigolizzo MB, Camilli JA, Francischone CE, Padovani CR, Brånemark P-I. Zygomatic bone: anatomic bases for osseointegrated implant anchorage. Int J Oral Maxillofac Implants. 2005;20:441–447. [Google Scholar]
- Rossi M, Duarte LR, Mendonça R, Fernandes A. Anatomical bases for the insertion of zygomatic implants. Clin Implant Dent Relat Res 2008;10:271–275. [Google Scholar]
- Bacci C, Manera C, Meneghetti D, Dhelpra I, Sbricoli L. Efficacy of Bichat’s buccal fat pad advancement in the treatment of odontogenic maxillary sinus fistulas. Surgeries 2025;6:10. [Google Scholar]
- Mohammadi A, Hassani A, Fazlisalehi O. Use of buccal fat pad in facial cosmetic surgery. In: Keyhan SO, Fattahi T, Bagheri SC, Bohluli B, Amirzade-Iranaq MH, eds Integrated Procedures in Facial Cosmetic Surgery, Cham: Springer International Publishing, 2021:887–901. [Google Scholar]
- Davis SJ, Park C, Shipchandler IT, Abdulhak A, Vernon D, Lee HB, et al. Buccal fat advancement-transposition flap for reconstruction of midface volume defects. Facial Plast Surg Aesthetic Med 2024;26:497–502. [Google Scholar]
- Akşamoğlu M, Bayar Muluk N, Şahan MH. Changes of the buccal fat pad volume according to the different age groups, gender, and body mass index: an evaluation with computed tomography. J Comput Assist Tomogr 2025;49:156–164. [Google Scholar]
- De Moraes E. The buccal fat pad flap: an option to prevent and treat complications regarding complex zygomatic implant surgery. preliminary report. Int J Oral Maxillofac Implants 2012;27:905–910. [Google Scholar]
- Abdel-Fatah R, Saleh W, El-Sharkawy H. Efficacy of buccal pad fat as a new approach in the treatment of gingival recession: a systematic review. BMC Oral Health 2024;24:768. [Google Scholar]
- Chrcanovic BR, Abreu MHNG. Survival and complications of zygomatic implants: a systematic review. Oral Maxillofac Surg 2013;17:81–93. [Google Scholar]
- Aparicio C, Manresa C, Francisco K, Claros P, Alández J, González‐Martín O, et al. Zygomatic implants: indications, techniques and outcomes, and the zygomatic success code. Periodontol 2000 2014;66:41–58. [Google Scholar]
- Aparicio C, Aparicio A. ZAGA double pedicle palatal flap for soft tissue management in zygomatic surgery. Int J Periodontics Restorative Dent 2024;45:1–23. [Google Scholar]
- Leroy C, Longis J, Bertin H, Mader M, Bonnet R. Complications and risk factors associated with zygomatic implants: retrospective analysis with 73 consecutive patients followed for 3.5 years. Quintessence Int (Berl) 2025;56:46. [Google Scholar]
- Lobna Elwan, Elprince N, Sweedan A. Evaluation of using buccal pad of free fat tissue transfer with immediate implant placement in the maxillary posterior area (clinical trial). Alexandria Dent J 2021;46:43–49. [Google Scholar]
- De Moraes EJ, Leigthon Y, Olate S, De Moraes LEB. The buccal fat pad technique an option to prevent complications in zygomatic implants surgery: 5–13 years follow up. SVOA Dent 2023;4:162–171. [Google Scholar]
- Samson J, Pandurangan P, Balasubramanian A, Jayabalan J. Versatility of buccal pad of fat in the reconstruction of oral and maxillofacial defects - a review. J Evol Med Dent Sci 2021; 10:3441–3445. [Google Scholar]
Cite this article as: Gautier G, Gosseiin-Rousselle L, Henry D, Salmon B, Moreau N. 2026. Iatrogenic osteomyelitis in patients with florid cemento-osseous dysplasia: an illustrative case report. J Oral Med Oral Surg. 32, 4. https://doi.org/10.1051/mbcb/2026006
All Tables
Comparison between the study groups regarding implant survival rate, PISTR, and PISTC
All Figures
 |
Fig. 1 Virtual preoperative planning for zygomatic implants using Blue Sky software. |
| In the text | |
 |
Fig. 2 Quad extra-maxillary zygomatic implants in situ, with healing abutments in place. |
| In the text | |
 |
Fig. 3 The pedicled buccal fat pad flap covered the zygomatic implants on one side and was sutured to the palatal mucosa with resorbable sutures. |
| In the text | |
 |
Fig. 4 Clinical photograph taken three months postoperatively illustrating the right side, where the BFPF was utilized to cover the EZI, demonstrating enhanced mucosal coverage and increased soft tissue thickness compared with the contralateral side (left), which exhibits visible gingival recession. |
| In the text | |
 |
Fig. 5 A six-month postoperative intraoral photograph shows sustained mucosal coverage and increased soft tissue thickness on the right side treated with the BFPF, contrasting with gingival recession on the left side. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


