| Issue |
J Oral Med Oral Surg
Volume 32, Number 1, 2026
|
|
|---|---|---|
| Article Number | 3 | |
| Number of page(s) | 5 | |
| DOI | https://doi.org/10.1051/mbcb/2026002 | |
| Published online | 17 March 2026 | |
Case Report
Primary intraosseous squamous cell carcinoma mimicking an odontogenic cyst: a case-report
Department of Oral Surgery, Center of Dental Care, Rennes, France, 2 Rue Henri le Guilloux cedex, 35000 Rennes
* Correspondence: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
2
July
2025
Accepted:
7
January
2026
Abstract
Introduction: A rare case of primary intraosseous squamous cell carcinoma (PIOSCC) emerging from an apical cyst is presented. Main symptoms and findings: An 80-year-old woman presented with an asymptomatic radiolucent lesion in the mandibular premolar region, identified during routine radiographic screening. The lesion was associated with chronic inflammation and prior endodontic treatment. Diagnoses, interventions, and outcomes: Initial management included enucleation of the lesion with extraction of the involved teeth. Histopathology revealed a moderately differentiated PIOSCC. Following oncological evaluation, the patient underwent curative surgery and was scheduled for adjuvant therapy. Conclusion: Although apical cysts are typically benign, rare malignant transformation underscores the necessity for histopathological analysis of all excised lesions. This highlights the importance of vigilant diagnostic and therapeutic approaches for seemingly benign lesions.
Key words: intraosseous squamous cell carcinoma / apical cyst / mandibular lesion / case report / odontogenic malignancy
© The authors, 2026
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Apical cysts are among the most common odontogenic lesions, often arising secondarily to chronic periapical inflammation. While these lesions are benign and responsive to conservative treatment, malignant transformation, although extremely rare, can occur [1]. The first case of primary intraosseous squamous cell carcinoma (PIOSCC) found in literature is reported by Loos in 1913 [2] and is usually related to chronic inflammation created by chronic tooth infections [1].
We present the case of an 80-year-old woman with a mandibular apical cyst, later diagnosed as PIOSCC. This case report has been written in accordance with the CARE (Case Report) guidelines.
Observation
The patient was an 80-year-old woman presenting with a history of coronary stent placement for ischemic cardiopathy, lower left lip in situ squamous cell carcinoma (ipsilateral to the PIOSCC) and a frigore face paralysis. She was treated with acetylsalicylic acid, bisoprolol, glyceryl trinitrate and atorvastatin, was a former smoker and had no allergies. A routine radiographic examination in August 2024 revealed a radiolucent lesion in the left mandibular premolar region (Fig. 1), involving the first and second left lower premolar teeth (20 and 21, using the ADA system). Both teeth had undergone endodontic treatment 10 yr earlier, but a recurrence of apical granuloma was noted on both teeth on the periapical radiograph from May 2024 (Fig. 2). The patient reported no pain, swelling, or any associated systemic symptom.
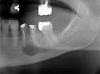 |
Fig. 1 Patient dental panoramic before primary attempt of apical cyst enucleation in august 2024. |
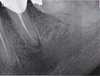 |
Fig. 2 Periapical radiograph (May 2024). |
Clinical findings
The extraoral examination revealed no visible lesions, no palpable mass, no signs of dysesthesia in the inferior alveolar nerve region, and no palpable lymphadenopathy.
Intraoral findings included a fluctuating swelling of the vestibule facing teeth 20 and 21, accompanied by a fistula that appeared only recently. The patient had missing teeth on both dental arches, and the mucosa looked healthy. Additionally, the teeth exhibited stage 2 mobility (Mühlemann 1954) and no response to percussion testing.
Diagnostic assessment
During the pre-operative consultation, the panoramic radiography (Fig. 1) revealed an unilocular radiolucent lesion with a major axis of more than 5 mm, located around the roots of teeth 20 and 21, extending toward the inferior alveolar nerve canal. Cone Beam Computed Tomography (CBCT) imaging showed a radiolucent and homogenous oval lesion with regular contours, measuring 10 × 12.7 × 7.5 mm, erasing the mental foramen and the cortical wall (Fig. 3).
Initial diagnostic hypotheses included an inflammatory apical cyst, a dentigerous cyst or an orthokeratinized cyst, indicating a simple enucleation with histopathological analysis.
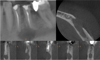 |
Fig. 3 CBCT sections showing clear cortical vestibular lysis; the letters F, L, and B indicate the Frontal, Lingual, and Buccal planes respectively. |
Therapeutic intervention
The initial procedure involved enucleation of the lesion and simultaneous extraction of teeth 20 and 21 under local anaesthesia. Complete excision was achieved despite the adherence of the lesion to the inferior alveolar nerve, and pathological analysis was requested.
Though seemingly benign, histopathological analysis revealed a well-defined cystic cavity, lined by a squamous epithelium that appeared locally close to normal, with progressive transformation into carcinoma in situ and then into invasive carcinoma (Fig. 4, no. 3). Indeed, histopathological examination reported a markedly thickened, stratified squamous epithelial lining exhibiting pronounced cytological atypia. Key features include nuclear pleomorphism, hyperchromatism, elevated nuclear-to-cytoplasmic ratios, and increased mitotic activity. Crucially, we observed focal breaches of the basement membrane with infiltration into the adjacent fibrous connective tissue. Although additional immunohistochemical markers such as Ki-67, p53, or cytokeratins were not deemed necessary given the typical histological features, PD-L1 immunostaining (clone 22C3, Dako) was performed for prognostic purposes and showed strong membranous positivity with a Combined Positive Score (CPS) of 100. Such an appearance may suggest malignant transformation of a cystic lesion. However, the possibility of a metastatic squamous cell carcinoma with a localized cystic appearance cannot be ruled out. Nevertheless, no synchronous squamous cell carcinoma was identified, which supports the hypothesis of malignant transformation arising from a pre-existing cystic lesion.
Cervical-facial CT scan and MRI identified a mandibular parasymphyseal osteolytic lesion confined to the jaw, and an invasion of the vestibular soft tissue with no evidence of adenopathy. According to the Zwetyenga CT scan classification for malignant intraosseous odontogenic tumors [3], this lesion was classified as stage T3.
An extension and exclusion assessment was carried out to rule out any distant primary tumors, including a PET scan that didn't reveal synchronic metastatic lesion.
Following staging assessment and multidisciplinary consultation meeting, and considering the patient's preserved general state, surgical management was proposed. Interruptive pelvi- mandibulectomy was performed along with left unilateral cervical lymph node dissection, and reconstruction was carried out using a fibula flap (Fig. 5). Surgical margins were confirmed as tumor- free, and no lymph node was invaded. Adjuvant radiotherapy was then completed (50 Gy on the tumor bed and on the lymph nodes).
The patient is being followed regularly with a focus on oral and dental maintenance to prevent osteoradionecrosis, comprising meticulous oral hygiene, topical fluoride application and conservative dental care instead of invasive procedures. The additional radiolucent areas identified on the panoramic radiograph are monitored clinically and radiographically as part of the follow-up care.
 |
Fig. 4 Three histological sections from the initial biopsy: No 1 (left) — Well-circumscribed cystic lesion located within the bone. H&E, original magnification × 20. No 2 (center) — Initial biopsy showing a well-differentiated invasive squamous cell carcinoma with a lobulated architecture. H&E, original magnification × 40. No 3 (right) — Same cystic lesion squamous epithelium transitioning from a normal appearance (left) to carcinoma in situ and then to invasive carcinoma with stromal invasion. H&E, original magnification × 200. |
 |
Fig. 5 Post reconstruction orthopantomogram with fibula flap. |
Discussion
The development of PIOSCC and its progression from odontogenic cysts, including apical cysts and odontogenic keratocysts (OKCs), are not yet fully understood. This condition is rare, as it represents 1 to 2% of all oral cancers [4]. PIOSCC predominantly affects males, with a male-to- female ratio of 2.22:1 and primarily occurs in adults between their 6th and 8th decades of life. It most frequently arises in the mandible (79%) and is primarily associated with residual or radicular cysts [4–6].
The pathogenesis of PIOSCC remains incompletely understood. However, chronic inflammation appears to be a critical factor driving malignant transformation. Persistent inflammatory stimuli can lead to epithelial dysplasia through DNA damage and reparative proliferation [7,8]. Odontogenic cysts, particularly apical cysts and OKCs, are potential sources of such chronic inflammation. Although parakeratinized OKCs are known for their aggressive nature and high recurrence rates, even orthokeratinized variants — previously considered less aggressive — have shown potential for malignant transformation [9–12]. These findings underline the importance of routine histopathological evaluation of all odontogenic cysts, especially in recurrent or atypical cases.
The diagnosis of PIOSCC requires strict criteria: the lesion must be intraosseous without initial mucosal or cutaneous involvement, no evidence of another primary tumor must be present at the time of diagnosis, and the histological type must be squamous cell carcinoma. PIOSCC is classified histologically into four subtypes: type 1 (associated with an odontogenic cyst), type 2 (malignant ameloblastoma or de novo ameloblastic carcinoma), type 3 (de novo intraosseous carcinoma), and type 4 (mucinous squamous cell carcinoma) [13,14]. Our case was most consistent with type 1 PIOSCC evolving from an apical cyst, the most common variant (Fig. 4).
Clinically, PIOSCC often follows an indolent course, with symptoms such as swelling, pain, or paraesthesia appearing late in the disease process. The literature reports a mean clinical duration of 5 yr before diagnosis in some cases [5]. Neurological symptoms, particularly paraesthesia of the lower lip, may indicate perineural invasion and should raise concern for malignancy. In our case, the lesion was initially asymptomatic and presumed benign, emphasizing the diagnostic challenge and need for clinical vigilance when evaluating longstanding or atypical cystic lesions through thorough radiographic description.
Radiographically, PIOSCC often mimics benign odontogenic cysts, presenting as unilocular or multilocular radiolucencies [15,16]. However, CT or CBCT can reveal features suggestive of malignancy, such as cortical plate perforation, ill-defined borders, or bone destruction. Histological confirmation remains essential and should demonstrate a transition from benign cystic epithelium to malignant epithelium, often supported by immunohistochemical analysis [4].
Recurrent cysts carry an increased risk of malignant transformation. OKCs, for instance, have a recurrence rate of up to 62% after surgical treatment. Factors contributing to recurrence include lesion size, location, inadequate primary treatment, and syndromic associations such as naevoid basal cell carcinoma syndrome. Recurrence may promote persistent inflammation, further increasing the risk of dysplastic progression [17].
From a radiological perspective, intraosseous tumors like PIOSCC should not be automatically staged as T4 based solely on their intraosseous location, as proposed by Zwetyenga et al. This misclassification could overstate the severity of disease and misguide treatment decisions [3].
Management of PIOSCC relies on a multidisciplinary approach. Surgical resection with negative margins remains the cornerstone, while radiotherapy is indicated in cases with positive margins or local recurrence. Neck dissection and chemotherapy are typically reserved for patients with clinical or radiological evidence of lymphatic or distant metastasis [12,18]. Interestingly, type 1 PIOSCC exhibits a relatively low cervical metastasis rate (10.3%) compared to types 2–4 (ranging from 36.5% to 70.1%), allowing for potentially more conservative nodal management. To our knowledge, no published case reports or clinical series describe treating primary intraosseous squamous cell carcinoma (PIOSCC) of the jaws with PD-1/PD-L1 checkpoint inhibitors. Although pembrolizumab and nivolumab are effective for metastatic head and neck squamous cell carcinoma, these results may not apply to PIOSCC. As a result, the prognostic or therapeutic relevance of PD-L1 expression in PIOSCC remains unclear and warrants further study.
Prognostically, type 1 PIOSCC demonstrates better outcomes, with a 5 yr survival rate of approximately 53.2%, compared to 36.3–38.8% for other types [4,12,19]. Tumors located in the maxilla also appear to have a more favorable prognosis than those in the mandible [6]. Local recurrence is the most significant negative prognostic factor and has been closely associated with reduced survival rates [20]. Thus, aggressive initial treatment, including radical resection and adjuvant radiotherapy when indicated, is crucial to improve long-term outcomes.
The patient was highly satisfied with the care provided by our teams, which was delivered in full accordance with current clinical guidelines and best practices.
Conclusion
PIOSCC arising from odontogenic cysts, although rare, poses significant diagnostic and therapeutic challenges. This case highlights the ongoing need for clinical vigilance and imaging in the evaluation of cystic lesions, particularly those with a prolonged clinical course or recurrent nature. Furthermore, routine histopathological evaluation and the critical role of multidisciplinary care are essential in managing such complex conditions. Large-scale studies are needed to further refine diagnostic and prognostic models for PIOSCC.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Conflicts of interest
The authors declare no conflicts of interest related to this work.
Data availability statement
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Ethics approval
This study is a single-patient case report based on standard clinical care and did not involve any experimental interventions or prospective data collection. In accordance with Rennes hospital policy and relevant national guidelines, ethics committee approval was not required.
Informed consent
Written Informed consent was obtained from the patient.
References
- Wang LL, Olmo H. Odontogenic Cysts 2022 Sep 26. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 34662043. [Google Scholar]
- D L. Central epidermoid carcinoma of the jaws. Dtsch Monatschr Zahnheilk 1913;31:308. [Google Scholar]
- Zwetyenga N, Pinsolle J, Rivel J, Majoufre-Lefebvre C, Faucher A, Pinsolle V. Primary intraosseous carcinoma of the jaws. Arch Otolaryngol Head Neck Surg 2001;127:794–797. [Google Scholar]
- Bodner L, Manor E, Shear M, van der Waal I. Primary intraosseous squamous cell carcinoma arising in an odontogenic cyst: a clinicopathologic analysis of 116 reported cases. J Oral Pathol Med Off Publ Int Assoc Oral Pathol Am Acad Oral Pathol 2011;40:733–738. [Google Scholar]
- Lugakingira M, Pytynia K, Kolokythas A, Miloro M. Primary intraosseous carcinoma of the mandible: case report and review of the literature. J Oral Maxillofac Surg 2010;68:2623–2629. [Google Scholar]
- de Morais EF, Carlan LM, de Farias Morais HG, Pinheiro JC, Martins HDD, Barboza CAG, et al. Primary intraosseous squamous cell carcinoma involving the jaw bones: a systematic review and update. Head Neck Pathol 2021;15:608–616. [Google Scholar]
- McGowan RH. Primary intra-alveolar carcinoma. A difficult diagnosis. Br J Oral Surg 1980;18:259–265. [Google Scholar]
- Jain M, Mittal S, Gupta DK. Primary intraosseous squamous cell carcinoma arising in odontogenic cysts: an insight in pathogenesis. J Oral Maxillofac Surg Off J Am Assoc Oral Maxillofac Surg 2013;71:e7–e14. [Google Scholar]
- Van der Waal I, Rauhamaa R, Van der Kwast WA, Snow GB. Squamous cell carcinoma arising in the lining of odontogenic cysts. Report of 5 cases. Int J Oral Surg 1985 Apr;14:146–152. [Google Scholar]
- Ward TG, Cohen B. Squamous carcinoma in a mandibular cyst. Br J Oral Surg 1963;1:8–12. [Google Scholar]
- MacLeod RI, Soames JV. Squamous cell carcinoma arising in an odontogenic keratocyst. Br J Oral Maxillofac Surg 1988;26:52–57. [Google Scholar]
- Ye P, Wei T, Gao Y, Zhang W, Peng X. Primary intraosseous squamous cell carcinoma arising from an odontogenic keratocyst: case series and literature review. Med Oral Patol Oral Cirugia Bucal 2021;26:e49–e55. [Google Scholar]
- Thomas G, Sreelatha KT, Balan A, Ambika K. Primary intraosseous carcinoma of the mandible—a case report and review of the literature. Eur J Surg Oncol EJSO 2000;26:82–86. [Google Scholar]
- Ray AC, Foletti JM, Graillon N, Guyot L, Chossegros C. Carcinomes intra-osseux primitifs des mâchoires de novo (type 3). Rev Stomatol Chir Maxillo-Faciale Chir Orale 2016;117:411–420. [Google Scholar]
- Yang CF, Tseng CH, Chen YK, Chen CM. Primary intraosseous carcinoma of the mandible. J Dent Sci 2020;15:236–238. [Google Scholar]
- Nomura T, Monobe H, Tamaruya N, Kishishita S, Saito K, Miyamoto R, et al. Primary intraosseous squamous cell carcinoma of the jaw: two new cases and review of the literature. Eur Arch Otorhinolaryngol 2013;270:375–379. [Google Scholar]
- Fidele NB, Yueyu Z, Zhao Y, Tianfu W, Liu J, Sun Y, et al. Recurrence of odontogenic keratocysts and possible prognostic factors: review of 455 patients. Med Oral Patol Oral Cir Bucal 2019 24:e491–e501. [Google Scholar]
- Boni P, Sozzi D, Novelli G, Pagni F, Valente G, Bozzetti A. Primary intraosseous squamous cell carcinoma of the jaws: 6 new cases, experience, and literature comparison. J Oral Maxillofac Surg 2016;74:541–546. [Google Scholar]
- Wenguang X, Hao S, Xiaofeng Q, Zhiyong W, Yufeng W, Qingang H, et al. Prognostic factors of primary intraosseous squamous cell carcinoma (PIOSCC): a retrospective review. PLOS ONE 2016 Apr 13;11:e0153646. [Google Scholar]
- Dujoncquoy JP, Rojare C, Chemli H, Wojcik T, Raoul G, Ferri J. Carcinome intra-osseux primitif des maxillaires traités par exérèse large et radiothérapie. Rev Stomatol Chir Maxillo- Faciale Chir Orale 2013;114:349–355. [Google Scholar]
Cite this article as: Barsacq A, Natu M, Hamon J, Guégan B, Dequidt A, Le Gall F, Zyani K, Lejeune S. 2026. Primary intraosseous squamous cell carcinoma mimicking an odontogenic cyst: a case-report. J Oral Med Oral Surg. 32: 3. https://doi.org/10.1051/mbcb/2026002
All Figures
 |
Fig. 1 Patient dental panoramic before primary attempt of apical cyst enucleation in august 2024. |
| In the text | |
 |
Fig. 2 Periapical radiograph (May 2024). |
| In the text | |
 |
Fig. 3 CBCT sections showing clear cortical vestibular lysis; the letters F, L, and B indicate the Frontal, Lingual, and Buccal planes respectively. |
| In the text | |
 |
Fig. 4 Three histological sections from the initial biopsy: No 1 (left) — Well-circumscribed cystic lesion located within the bone. H&E, original magnification × 20. No 2 (center) — Initial biopsy showing a well-differentiated invasive squamous cell carcinoma with a lobulated architecture. H&E, original magnification × 40. No 3 (right) — Same cystic lesion squamous epithelium transitioning from a normal appearance (left) to carcinoma in situ and then to invasive carcinoma with stromal invasion. H&E, original magnification × 200. |
| In the text | |
 |
Fig. 5 Post reconstruction orthopantomogram with fibula flap. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


