| Issue |
J Oral Med Oral Surg
Volume 30, Number 1, 2024
|
|
|---|---|---|
| Article Number | 6 | |
| Number of page(s) | 8 | |
| DOI | https://doi.org/10.1051/mbcb/2024009 | |
| Published online | 29 April 2024 | |
Original Research Article
Predictors of fistula formation after primary palatoplasty
Department of Oral & Maxillofacial Surgery, Faculty of Dentistry, University of Port Harcourt, Port Harcourt, Rivers State, Nigeria
* Correspondence: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
22
October
2023
Accepted:
11
March
2024
Abstract
Background: The goal of cleft palate repair is to create a seal between the oral and nasal cavities while improving speech by creating a functional velum. Various surgical techniques are at the surgeon's disposal to achieve this. Unfortunately, this sometimes fails, leading to formation of oronasal fistula. Objective: This study aimed to determine the predictors of oronasal fistula (ONF) formation following primary cleft palate repair. Methodology: This prospective interventional study involved cleft palate patients recruited from the University of Port Harcourt Teaching Hospital. Basic demographics and clinical data of participants were recorded preoperatively while morphometric measurements were taken under general anaesthesia to determine Cleft Palate Index and Cleft width. Four surgeons performed the procedures, and three surgical techniques were employed. Participants were followed up for a mean period of 5 months. The occurrence, location, and fate of ONF were documented. Correlation between ONF and potential predictor variables was tested, and determinant(s) of fistula formation was explored. Data was analysed using IBM SPSS version 21.0 with statistical significance set at P < 0.05. Result: Eighty-two participants with ages ranging from 9 months to 19 years completed the study. The initial incidence of ONF was 29.3% but 16.7% of these cases resolved spontaneously within 5 weeks. Cleft Palate Index, Cleft width, and time (age) of repair significantly correlated with occurrence of ONF. However, Cleft width was the only statistically significant (p = 0.049) individual determinant of fistula formation. Conclusion: In this study, Cleft width W is the most important predictor of ONF formation.
Key words: Cleft palate / surgery / fistula
© The authors, 2024
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
The goals of management of children born with cleft palate include creation of intact primary and secondary palate to achieve oral and nasal seals for the purpose of feeding and growth [1].
The incidence of post repair oronasal fistulae (ONF) has been reported to range from 5% to 60% [2]. The severity of the cleft, technique of surgical repair, timing of the repair, postoperative infection and experience of the surgeon are some of the fundamental factors associated with ONF formation. Once it occurs, oronasal fistula can be troublesome, both to patients and surgeons. Although, a high initial success rate of up to 85% of secondary oronasal fistula repair has been reported, a recurrence rate of 33–37% followed; hence, ONF remains a challenging problem [3]. Therefore, more studies are required to affirm the factors predictive of fistula formation to facilitate more effective case-by-case surgical planning. This study was intended to determine the effect of cleft severity and some other operative variables on the occurrence of ONF after primary palatoplasty.
Materials and methods
A prospective interventional study was conducted at the University of Port Harcourt Teaching Hospital [UPTH] Rivers State, Nigeria from October 2018 to May 2020 to elicit factors predictive of oronasal fistula formation after primary palatoplasty. Participants were selected from patients seeking treatment for congenital clefts of the secondary palate at the Oral and Maxillofacial Surgery and Plastic Surgery outpatient clinics. The inclusion criteria were non-syndromic cases of unilateral or bilateral cleft of the primary and/or secondary palate, “Age-Weight-Haemoglobin concentration” of 9 months, >10th percentile of expected weight for age, and 10 g/dl respectively, and no previous repair of the cleft palate. Syndromic cases were excluded because some often present with non-modifiable conditions that may hamper wound healing or predispose to infection. Similarly, extremely wide clefts were excluded as these often require modification of techniques with 2-stage closure, buccal myomucosal flap or FAMM flap etc. Preoperatively, active or passive palatal plates were not used in these group of patients. Written informed consents were obtained from adult subjects or parents/guardians of minor subjects before enrolment in the study. Funding of treatments and care was covered under the subsisting Smile Train grant at the Hospital. Approval of the study protocol was obtained from the Institutional Research and Ethics Committee.
Data collection
Preoperatively, a data sheet was used to record patient's demographic details and relevant clinical data including: sex, weight, cleft type (Veau's classification) and time (age) of repair (ToR). The time of repair was defined by developmental bracket described by Erickson [4] and derived based on patient's age. This includes infant (<2 years), toddler (>2–3 years), Preschooler (>3–5 years), School age (6–12 years), and Adolescent (12–19 years). Morphometric measurements of the cleft palate were taken after intubation and induction of general anaesthesia. Cleft severity was determined based on the Cleft Palate Index calculated from some of the measurements taken.
Morphometric measurements of the cleft palate
Under general anaesthesia, Boyle-Davis or Dingman's retractor was inserted to expose the palatal defect by ensuring visibility and accessibility from the uvula to the anterior maxillary alveolus. In clefts involving both hard and soft palate, using a Castroviejo calliper, the Cleft width (W) was measured along the junction of the hard and soft palate. This was the distance between the cleft margins. Next, the palatal segments on either side of the cleft defect were measured. This was the distance between cleft margin and the gingival margin taken at the same level as the Cleft width (Fig. 1). For isolated clefts of the soft palate, the distance between the base of the cleft uvula (Uvular gap) on either side was taken as the Cleft width (W) while the palatal segment on either side was measured from the cleft margin perpendicularly to the marked line of linear incision extending from mid maxillary tuberosity posteriorly across the mucous membrane over the hamular notch. Measurements taken from the right and left sides were designated as P1 and P2 respectively. The Cleft Palate Index (CPI) was calculated by dividing the Cleft width (W) by the sum of the two palatal segments 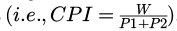 Based on the CPI, severity of cleft deformity was classified according to Rossell-Perry's criteria [5,6] stated as: Mild (CPI < 0.2), Moderate (CPI = 0.2–0.4), Severe (CPI > 0.4). Notably however, patients with extremely wide clefts were excluded from this study, as defined by Bumsted [7]; that is: “where the width of the cleft defect is larger than the sum of the combined widths of the remaining palatal segments.
Based on the CPI, severity of cleft deformity was classified according to Rossell-Perry's criteria [5,6] stated as: Mild (CPI < 0.2), Moderate (CPI = 0.2–0.4), Severe (CPI > 0.4). Notably however, patients with extremely wide clefts were excluded from this study, as defined by Bumsted [7]; that is: “where the width of the cleft defect is larger than the sum of the combined widths of the remaining palatal segments.
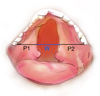 |
Fig. 1 Illustration of morphometric measurements. |
Surgical procedure
Patients were randomly assigned to four very experienced cleft surgeons. Intravenous ceftriaxone (50 mg/kg or 1 g for adults) was administered at induction of anaesthesia. Palatoplasties were accomplished by either von Langenbeck's, Bardach's or intravelar veloplasty techniques. Generally, all isolated clefts of the soft palate had intravelar veloplasty technique alone, complete cleft of the secondary palate had von langenbeck repairs with intravelar veloplasty while complete cleft of the primary and secondary palate had Bardach's repair with intravelar veloplasty except in few cases for the reason of surgeon's personal preference. Vomerine flaps were adopted to complete the closure of the nasal layer in some cases of complete cleft of the primary and hard palate.
Postoperative care
Syringe bulb feeding with clear fluid diet commenced after full post anaesthesia recovery usually not later than 6 hours post op. Arm restraints were used in the paediatric group for 72 hours following surgery. Cup-and-spoon-feeding commenced 48 hours post op. Active sucking by infants was not allowed until 3 weeks post op. Semi solid diet [pap, custard] commenced by the second day post op. Nurse/parent − assisted oral toileting with warm saline six times daily which commenced 24 hours post op. Patients were discharged home 48 hours post op after thorough inspection of surgical sites. Oral toileting instructions were reiterated with emphasis at discharge. Patients were placed on calculated doses of analgesics (NSAIDs), anti-inflammatories (Steroids) and antibiotics (Penicillins) for 6 days.
Postoperative review and outcome recording
Participants were reviewed weekly at the Oral and Maxillofacial Surgery outpatient clinic for the first 5 weeks, and monthly afterwards. At each visit, participants were examined by the principal investigator (KSY − first author) and verified by any one of the other three surgeons in order to document the status of surgical wound healing, presence or absence of a fistula, number of fistulae, location of fistula and ultimately, the fate of the fistula (i.e., healed secondarily or requiring re-operation). A fistula was deemed to have occurred if an obvious oronasal communication was observed anywhere along the line of repair; especially when associated with nasal regurgitation and/or hypernasality.
Data analyses
Data collected was entered into a computer spreadsheet and analyzed using the SPSS Version 21.0 for IBM Corp, USA. First, the association of cleft severity on ONF formation was assessed using Fisher's exact test. Afterwards, bivariate Pearson's test was conducted to determine the correlation between occurrence of ONF and morphometric variables measured in continuous numerical values such as CPI, W, P1 + P2. In the same vein, correlation of ONF to qualitative variables such as cleft type, surgical technique of repair, patient's weight and time (age) of repair was assessed with Spearman's rho correlation test. Variables with significant correlation coefficients from these initial correlation analyses were adopted as covariates in a logistic regression model to establish the true determinants of ONF occurrence. Statistical significance was established at 95% confidence interval with p-values < 0.05 in all cases.
Results
Eighty-two participants completed the study, including 38 (46.3%) males and 44 (53.7%) females; giving a male to female ratio of approximately 1:1.2. They weighed between 7kg and 62 kg. Participants' ages ranged from 9 months to 19 years with a mean value of 6.12 ± 5.88 years. Age classification according to developmental milestones showed predominance of repairs at infancy (40.0%) and adolescence (20.0%). Categorical variables are shown in Table I: In terms of cleft severity, most cases (54, 65.9%) were of moderate severity while mild and severe cases were equal in frequency. Veau's type II and III were jointly most frequent with 26 cases each, and the three surgical techniques used were fairly evenly employed. For the morphometric data, Cleft width (W) ranged from 5 to 22 mm with a mean of 10.8 mm; combined palatal tissue segments (P1 +P2) ranged from 23 to 51 mm with a mean value of 37.6 mm; while calculated Cleft Palate Index (CPI) ranged from 0.11 to 0.56 with a mean of 0.29.
Postoperative fistula occurred in 24 (29.3%) patients, and it mostly occurred in a single site (19, 79.2%) and predominantly at the junction of the soft and hard palate (20 cases). While 5 (20.8%) participants had fistulae at multiple sites. Most fistulae (16, 66.7%) occurred within the first week of the repair while the others (8, 33.3%) occurred by the second week. There were 14 cases of fistulae associated with moderately severe clefts, constituting 25.9% of participants in the group compared to 10 cases of fistulae constituting 71.4% of participants with severe clefts; no patient with cleft of mild severity developed a fistula. Spontaneous closure was observed in 4 (16.7%) out of the 24 cases: 2 each by the 4th and 5th postoperative week.
Fisher's exact test was conducted to assess the statistical difference between the various degrees of cleft severity in terms of their association with ONF after primary palatoplasty. (Tab. II). The observed likelihood ratio (20.586) and Fisher exact (17.646) values were highly significant (p = 0.000). Correlation between the morphometric parameters such as Cleft width (W), Cleft Palate Index (CPI), Palatal tissue segments (P1+P2) and occurrence of fistula was determined using Pearson's test (Tab. III) while correlation between the categorical variables (Cleft type, Cleft severity, time (age) at repair, surgical technique) was determined with Spearman's rho test (Tab. IV) .There were significant correlations between occurrence of fistula and Cleft width (W), Cleft Palate Index (CPI) and time (age) of repair (Tab. V). However, the correlation coefficient for Cleft width (r = 0.511) was marginally higher than that of Cleft Palate Index (r = 0.456). The minimum Cleft Palate Index at which fistula occurred was 0.22.
To unveil the actual determinant(s) of fistula formation, a binary logistic regression model was developed with the three significant correlates i.e., Cleft width, Cleft Palate Index, and time (age) of repair (ToR), as independent covariates determining the occurrence or non-occurrence of ONF (dependent variable) after primary palatoplasty. The relative predictive values of the individual covariates as depicted on Table V, shows that Cleft width (W) was the only significant (p = 0.049) individual determinant of fistula formation.
Frequency distribution of categorical variables.
Cleft severity and association with occurrence of oronasal fistula (ONF).
Correlation of morphometric parameters with occurrence of ONF.
Correlation of categorical variables with Occurrence of ONF.
Relative predictive values of covariates in the logistic regression model.
Discussion
Oronasal fistula formation is perhaps the most perplexing complication of primary palatoplasty. It is therefore, essential to determine and mitigate the causative factors as much as possible. In this study, the final incidence of persistent ONF was 24.3%, which falls within the world-wide range of between 5% and 60% [2,8] reported in the literature. Although far higher in comparison to 9.94% reported in a systematic review by Tache et al. [9] and 4.9% reported in a meta-analysis by Bykowsky et al. [10]. It must be noted the latter figures were mean rate derived from only 11 articles and 35 articles respectively, while several other individual studies had reported much higher fistula rates. Daniel et al. [11] established a relativity between fistula formation in cleft palate patients treated in low resource economy and on humanitarian ground compared to those treated in established tertiary institutions and resource endowed society. The key influential variables associated with higher fistula rate in the poor resource and humanitarian situation (35.5% − ReSurge International) compared to tertiary institutions and rich economy patients (12.8% − Huaxi University, China; 2.5% − University of Southern California, US) were older age, and more severe Veau's classifications (III or IV). While majority of patients treated in tertiary hospitals and rich economy were less than 2 years and of Veau's classes I or II, those managed in poor resource by humanitarian intervention were predominantly older than 2 years and of Veau's classes III or IV. These characteristics were similarly observed in the patients involved in our study with a comparable population of infants and adolescents, a comparable Veau III or IV versus Veau I or II population, and mostly beneficiaries of humanitarian sponsorship by the Smile Train organisation through a free surgical outreach conducted in our hospital. Daniel et al.'s hypothesis of impact of poor resource economy on post-palatoplasty fistula rates therefore holds in this situation. It would appear that patients' present late, when older in resource poor settings. As they often must await free or opportunities for humanitarian intervention since many cannot afford the cost of treatment by themselves. At such time, the cleft width may have become wider.
It is noteworthy, however, that spontaneous closure of fistula occurred in 16.7% of the cases in the present study before or by the 5th postoperative week while 83.3% persisted until the 5th month and would require secondary closure. Hence, close follow up of a dehiscent repair is advisable; at least for one month, since spontaneous closure has also been reported by other authors such as Lu et al. [12] who similarly observed closure in 4 patients within 4 weeks.
In consistency with established knowledge, single site fistulation was considerably more common and generally, fistulae most frequently occurred at the junction of the hard and soft palate in this study. Nonetheless, multiple fistulae are not uncommon; there were five cases constituting 20.8% of the occurrences in this study. Coincidentally, Passos et al. [13] had also reported five cases of multiple fistulae, though, in a much larger sample − 589 cases of palatal cleft repairs. It is noteworthy that none of the cases with multiple fistulae resolved by spontaneous closure in the present study. The propensity for dehiscence at the junction of the hard and soft palate has largely been imputed to the facts that palatal cleft is usually widest in this region, as well as mucosa atrophy, inadequacy of muscle bulk and insufficient muscle mobilization for closure at that central area, resulting in greatest tension at closure [13,14], coupled with constant motion of the soft palate as it hinges on the hard palate during velopharyngeal function [15].
Morphological severity of the palatal cleft has been presumed responsible for ONF after palatoplasty. However, the definition of severity in this context, has been elusive. Older studies defined severity according to Veau's classification of the cleft deformity, whereas, newer studies assigned severity based on Cleft width or Cleft Palate Index. All these parameters were assessed in this study in order to determine their relative impact and potential causality of ONF after primary palatal cleft repairs. In addition, other demographic and operative variables such as developmental age at the time of repair, palatal tissue segment, patient's weight and surgical technique were also interrogated to elicit the correlates and determinants of ONF occurrences.
To start with, this study confirmed strong association between cleft severity; defined by Cleft Palate Index (CPI), and occurrence of ONF, with very strong likelihood as the severity increases from moderate to severe. This finding was further reinforced by the significant positive correlation observed between Cleft Palate Index and occurrence of ONF. On the contrary, Veau's classification did not demonstrate strong correlation with ONF formation in this study. This gives credence to the more recent approach whereby; severity is defined more by CPI rather than the older methods which used Veau's classification. Park et al. [16] similarly noted that Veau classification was not significant in their study. Although Yuan et al. [17] particularly noted Veau Class IV to be associated with ONF and Daniel et al. [11] observed association with Veau Class III or IV. On the contrary, Nozomi Tosa et al. [18] recorded no fistula despite having comparable proportion of Veau Class IV patients in their study population. The proportion of Veau Class IV patients in our study is considerably small compared to other classes, this may partly account for its lack of significance in this study. In fact, Bykowski et al. [10] had noted that significant relationship between ONF and Veau's classification is often observed when analysing large cohort; our study comprised only a small cohort of 82 patients; this may also contribute to the non-significance of Veau's classification.
Cleft width was another morphometric correlate of ONF occurrence while palatal tissue segment was not significantly so. Rossel-Perry et al. [6] in 2014, had reported the association between Cleft Palate Index, velopharyngeal insufficiency (VPI) and oronasal fistula (ONF) formation after palatoplasty, this observation is now being corroborated by our own finding. There was a higher fistula rate in patients with severe clefts. Similarly, Amirize et al. [19] in 2017, also confirmed an association between Cleft Palate Index and fistula occurrence, asserting that CPI in excess of 0.26 significantly predisposed to ONF. CPI calculation entails variables such as Cleft width and palatal tissue deficiency which are more pertinent than mere morpho-anatomical appearance which underpins Veau's classification.
Some more recent reports [14,20–24] are emphasizing on Cleft width as an isolated, singular entity critical to fistula formation after palatal cleft repair. In consonance with this assertion, this present study established significant correlation between Cleft width and ONF occurrence which was marginally superior to that demonstrated by CPI. Parwaz et al. [20] signified Cleft width >15 mm as a major predictor of fistula formation, a predictive value comparable to >13mm reported by Landheer et al. [24] in a later study. On the contrary, Yuan et al. [17]‘ found no association between Cleft width, as an isolated entity, and fistula formation. However, they noted that Cleft width was significantly associated with velopharyngeal insufficiency. In this present study, Cleft width was not only the strongest correlate of ONF occurrence, it, in fact, proved to be the singular most important determinant of fistula formation after primary repair. This finding agreed with Katusabe et et al.'s [25] who also reported that Cleft width was an independent predictor of palatal fistula following a multivariate analysis.
Although both CPI and Cleft width showed significant correlation to ONF occurrence, it was cleft width that showed the greater correlation coefficient suggesting that it is a stronger predictor of occurrence than CPI. This picture was further reinforced by the result of the binary logistic regression (Tab. V) which indeed proved that Cleft width is in fact, the sole significant individual determinant of ONF occurrence in this study.
Patient's developmental age at the time of repair (coded ToR) was the only other variable that showed significant positive correlation to the occurrence of ONF in this study. Although this observation has been contradicted by a recent study [26] which observed that timing of surgery made no difference to fistula development, the authors' observation arose from a dichotomy of patients into age groups < 4 years vs >4 years. This categorisation is at variance with what we did in this study whereby patients were grouped by their developmental milestones into infant, toddler, preschooler, school age and adolescents. However, the impact of our observed correlation in this study requires further verification because oral activities of the patient at the respective stage of development, rather than the absolute number of their age or quality of tissue and repairs, may be more influential. Since there was no known precedence to this particular research interrogation, our observation can only be cautiously asserted. However, it is important to state that time of repair (ToR) was not a significant predictor or determinant of ONF formation, even in this study.
Furthermore, as a notable limitation to this study, whereas surgeon's experience and surgical technique are other variables that have been implicated for fistula development, none of these could be fairly assessed in this research. This is because the surgeons were largely of comparable practice experience in cleft surgery. Moreover, essentially, all cases involved intravelar veloplasty combined with a two-flap technique where indicated; and as determined by individual surgeon's judgement and preferences. Hence, the correlation of surgical technique with ONF outcome in this study was weak and insignificant. Park et al. [16] alluded to a greater tendency for Furlow's technique to result in ONF; Furlow technique was not at all used in this study. Although some authors [27,28] have alleged the propensity for maxillary growth restriction and ONF with single stage closure, equally, there are contradictory reports disclaiming the superiority of 2-stage repair [29,30]. A systemic review conducted by Reddyl et al. [31] compared one-stage and two stage repair and concluded that there is no conclusive evidence of a relationship between one- or two-stage palatoplasty and facial growth, speech and fistula formation in patients with unilateral cleft lip and palate. Nevertheless, we routinely follow up our cleft surgery patients after surgery and individuals requiring further intervention are selected for subsequent management.
Extremely wide clefts, as defined by Bumsted [5], were excluded from the present study. It has already been established that the two-flap techniques employed in this study may not achieve tension-free closure in extremely wide clefts. Though, adjunct procedures such as addition of pedicled buccal fat pad [32,33] or acellular dermal matrix may improve outcome of one stage palatoplasty [34,35]. In situations where extremely wide clefts are to be managed, procedures such as use of Facial Artery Myo Mucosal Flap (FAMM flap), islandized mucoperiosteal flap, and two-stage repair have also been employed [36–39].
Conclusion
Essentially, this study has corroborated earlier reports associating cleft severity with the occurrence of oronasal fistula (ONF). In this case, severity was defined by Cleft Palate Index which, in addition to Cleft width, significantly correlates with the likelihood for ONF formation. However, Cleft width is the singular most important predictor of fistula formation following primary palatoplasty.
Acknowledgements
My immense gratitude goes to Dr. Tope Osagbemiro, my revered statistician who assisted with Data analysis and Dr Iroro Yarhere for his role in proofreading the manuscript.
Funding
Funding for the participants' treatments was covered under the Smile Train grant which provides sponsorship for all congenital cleft lip and palate repair at the University of Port Harcourt Teaching Hospital. This research did not receive funding from any organization.
Conflicts of Interest
Authors have no conflict of interest to disclose.
Data availability statement
The data supporting the findings of this study are available from the corresponding author upon request.
Ethics approval
Approval of the study protocol was obtained from the Research and Ethics Committee of the University of Port Harcourt Teaching Hospital.
Informed consent
Written informed consents were obtained from patients or their parents/guardians before enrolment as participants in the study.
References
- Costello BJ, Ruiz RL. Cleft lip and palate: Comprehensive treatment planning and primary repair. In: Miloro M, Ghali GE, Larsen PE, Waite PD, editors. Peterson's principles of oral and maxillofacial surgery. Hamilton, London: DC Becker Inc; 2004. p. PL 839–58. [Google Scholar]
- Phua YS, de Chalain T. Incidence of oronasal fistulae and velopharyngeal insufficiency after cleft palate repair: an audit of 211 children born between 1990 and 2004. Cleft Palate Craniofac J 2008;45:172–178. [CrossRef] [PubMed] [Google Scholar]
- Diah E, Lo LJ, Yun C, Wang R, Wahyuni LK, Chen YR. Cleft oronasal fistula: a review of treatment results and a surgical management algorithm proposal. Chang Gung Med J 2007;30:529–537. [PubMed] [Google Scholar]
- Erikson EH. Childhood and society. 1st edn. WW Norton and Company; 1950 [Google Scholar]
- Rossell-Perry P. New diagram for cleft lip and palate description: the clock diagram. Cleft Palate Craniofac F J. 2009;46:305–313. [CrossRef] [PubMed] [Google Scholar]
- Rossell-Perry P, Caceres Nano E, Gavino-Gutierrez AM. Association between palatal index and cleft palate repair outcomes in patients with complete unilateral cleft lip and palate. JAMA Facial Plast Surg 2014;16:206–210. [CrossRef] [PubMed] [Google Scholar]
- Bumsted RM. A new method for achieving complete two-layer closure of a massive palatal cleft. Arch Otolaryngol 1982;108:147–150. [CrossRef] [Google Scholar]
- Saothonglang K, Punyavong P, Winaikosol K, Jenwitheesuk K, Surakunprapha P. Risk factors of fistula following primary palatoplasty. J Craniofac Surg 2021;32:587–590. [CrossRef] [PubMed] [Google Scholar]
- Tache A, Mommaerts MY. On the frequency of oronasal fistulation after primary cleft palate repair. Cleft Palate Craniofac J 2019;56:1302–1313. [CrossRef] [PubMed] [Google Scholar]
- Bykowski MR, Naran S, Winger DG, Losee JE. The rate of oronasal fistula following primary cleft palate surgery: a meta-analysis. Cleft Palate Craniofac J 2015;52:e81–87. [CrossRef] [Google Scholar]
- Daniels KM, Yang Yu E, Maine RG, Heng Y, Yang L, Shi B, et al. Palatal fistula risk after primary palatoplasty: a retrospective comparison of a humanitarian organization and tertiary hospitals. Cleft Palate Craniofac J 2018;55:807–813. [CrossRef] [PubMed] [Google Scholar]
- Lu Y, Shi B, Zheng Q, Hu Q, Wang Z. Incidence of palatal fistula after palatoplasty with levator veli palatini retropositioning according to Sommerlad. Br J Oral Maxillofac Surg 2010;48:637–640. [CrossRef] [PubMed] [Google Scholar]
- de Agostino Biella Passos V, de Carvallho Carrara CF, da Silva Dalben G, Costa B, Gomide MR. Prevalence, cause, and location of palatal fistula in operated complete unilateral cleft lip and palate: retrospective study. Cleft Palate Craniofac J 2014;51: 158–164. [CrossRef] [PubMed] [Google Scholar]
- Amirize EE. The relationship between cleft width and oronasal fistula. Nigerian J Plast Surg 2016;12:4–11. [CrossRef] [Google Scholar]
- Suzuki A, Mukai Y, Ohishi M, Miyanoshita Y, Tashiro H. Relationship between cleft severity and dentocraniofacial morphology in Japanese subjects with isolated cleft palate and complete unilateral cleft lip and palate. Cleft Palate Craniofac J 1993;30:175–181. [CrossRef] [PubMed] [Google Scholar]
- Park MS, Seo HJ, Bae YC. Incidence of fistula after primary cleft palate repair: a 25-year assessment of one surgeon's experience. Arch Plast Surg 2022;49:43–49. [CrossRef] [PubMed] [Google Scholar]
- Yuan N, Dorafshar AH, Follmar KE, Pendleton C, Ferguson K, Redett RJ 3rd. Effects of cleft width and veau type on incidence of palatal fistula and velopharyngeal insufficiency after cleft palate repair. Ann Plast Surg 2016;76:406–410. [Google Scholar]
- Nozomi T, Yasuyoshi T, Shinya Y, Haruhisa N, Vivier M, Tomoaki K, et al. Incidence and contributing factors for palatal fistula after primary palatoplasty in medical collaborative program in Madagascar. Showa Univ J Med Sci 2018;30:271–283. [CrossRef] [Google Scholar]
- Amirize EE, Onah II, Gbeneol TJ, Akadiri OA. The relationship between cleft palate index and oronasal fistula. J Cleft Lip Palate Craniofac Anomal 2017;4:160–166. [CrossRef] [Google Scholar]
- Parwaz MA, Sharma RK, Parashar A, Nanda V, Biswas G, Makkar S. Width of cleft palate and postoperative palatal fistula − do they correlate? J Plast Reconstr Aesthet Surg 2009;62:1559–1563. [CrossRef] [PubMed] [Google Scholar]
- Musgrave RH, Bremner JC. Complications of cleft palate surgery. Plast Reconstr Surg Transplant Bull. 1960;26:180–189. [CrossRef] [Google Scholar]
- Amaratunga NA. Occurrence of oronasal fistulas in operated cleft palate patients. J Oral Maxillofac Surg. 1988;46:834–838. [CrossRef] [Google Scholar]
- Schultz RC. Management and timing of cleft palate fistula repair. Plast Reconstr Surg 1986;78:739–745. [CrossRef] [PubMed] [Google Scholar]
- Landheer JA, Breugem CC, Van der Molen ABM. Fistula incidence and predictors of fistula occurrence after cleft palate repair: two-stage closure versus one-stage closure. Cleft Palate Craniofac J 2010;47:623–630. [CrossRef] [PubMed] [Google Scholar]
- Katusabe JL, Hodges A, Galiwango GW, Mulogo EM. Challenges to achieving low palatal fistula rates following primary cleft palate repair: experience of an institution in Uganda. BMC Res Notes 2018;11:358. [CrossRef] [PubMed] [Google Scholar]
- Opris DA, Opris H, Dinu C, Bran S, Baciut G, Armencea G, et al. Evaluation of prognostic factors for palatal fistulae after cleft lip and palate surgery in a North-Western Romanian population over a 10-year period. Int Environ Res Public Health 2021;18:7305. [CrossRef] [Google Scholar]
- Yamanishi T, Nishio J, Kohara H, Hirano Y, Sako M, Yamanishi Y, et al. Effect on maxillary arch development of early 2-stage palatoplasty by modified furlow technique and conventional 1-stage palatoplasty in children with complete unilateral cleft lip and palate. J Oral Maxillofac Surg 2009;67:2210–2216. [CrossRef] [PubMed] [Google Scholar]
- Fudalej PS, Wegrodzka E, Semb G, Hortis-Dzierzbicka MJ. One-stage (Warsaw) and two-stage (Oslo) repair of unilateral cleft lip and palate: craniofacial outcomes. J Cranio-maxillofac Surg 2015;43:1224–1231 [CrossRef] [Google Scholar]
- Kappen IFPM, Bittermann D, Janssen L, Bittermann GKP, Boonacker C, Haverkamp S, et al. Long-term follow-up study of young adults treated for unilateral complete cleft lip, alveolus, and palate by a treatment protocol including two-stage palatoplasty: speech outcomes. Arch Plast Surg 2017;44:202–229. [CrossRef] [PubMed] [Google Scholar]
- Holland S, Gabbay JS, Heller JB, O'Hara C, Hurwitz D, Ford MD, et al. Delayed closure of the hard palate leads to speech problems and deleterious maxillary growth. Plast Reconstr Surg 2007;119:1302–1310. [CrossRef] [PubMed] [Google Scholar]
- Reddy RR, Reddy SG, Vaidhyanathan A, Berge SJ, Kuijpers-Jagtman A. Maxillofacial growth and speech outcome after one-stage or two-stage palatoplasty in unilateral cleft lip and palate: a systematic review. J Cranio-maxillofac Surg 2017;45:995–1003 [CrossRef] [Google Scholar]
- Ganatra MA. Use of buccal fat pad flap in palate repair. In: Fayyaz GQ (eds.), Surgical Atlas of Cleft Palate and Palatal Fistulae. Singapore: Springer 2022;359–364. [CrossRef] [Google Scholar]
- Adeyemo WL, Ibikunle AA, James O, Taiwo OA. Buccal fat pad: a useful adjunct flap in cleft palate repair. J Maxillofac Oral Surg 2019;18:40–45. [CrossRef] [PubMed] [Google Scholar]
- Aldekhayel SA, Sinno H, Gilardino MS. Acellular dermal matrix in cleft palate repair: an evidence-based review. Plast Reconstr Surg 2012;130:177–182. [CrossRef] [PubMed] [Google Scholar]
- Kattan AE., Abdulghafour M., Ahmed BA., Gelidan AG., Alhumsi TR. The use of acellular dermal matrix in palatoplasty to decrease the rate of postoperative oronasal fistula. J Pak Med Assoc 2022;72:337–341. [PubMed] [Google Scholar]
- Gupta R. Buccinator myomucosal flap in cleft palate repair: revisited. J Cleft Lip Palate Craniofac Anomal 2014;1:17–18. [CrossRef] [Google Scholar]
- Aboul-Wafa AM. Islandized mucoperiosteal flaps: a versatile technique for closure of a wide palatal cleft. Can J Plast Surg 2012;20:173–177. [CrossRef] [PubMed] [Google Scholar]
- Kreshanti P, Sadikin PM, Anggraeni MI, Wibowo JA, Bangun K. Surgical techniques to reduce oronasal fistula risk in wide cleft palate repair: a systematic review. Med J Indones 2022;31:250–259. [Google Scholar]
- Mat Saad AZ, Chai KS, Wan Sulaiman WA, Mat Johar SFN, H alim AS. Early outcomes of cleft and palatal width following anterior palate repair (vomerine flap) in infants with wide cleft lip and palate. Arch Plast Surg 2019;46:518–524. [CrossRef] [PubMed] [Google Scholar]
All Tables
All Figures
 |
Fig. 1 Illustration of morphometric measurements. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


