| Issue |
J Oral Med Oral Surg
Volume 29, Number 3, 2023
|
|
|---|---|---|
| Article Number | 28 | |
| Number of page(s) | 9 | |
| DOI | https://doi.org/10.1051/mbcb/2023025 | |
| Published online | 31 August 2023 | |
Educational Article
Management of biologics in oral surgery
Department of Oral and Maxillofacial Surgery, Norfolk and Norwich University Hospital, UK
* Correspondence : This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
22
April
2023
Accepted:
11
July
2023
Abstract
Biologics have revolutionised the management of immune-mediated inflammatory disorders, and their growing use has resulted in increasing numbers of these patients presenting in oral surgery settings. While the efficacy of biologics has been well established, they are associated with numerous adverse effects. Several professional bodies have published recommendations on perioperative management of these agents to reduce complications. Currently no specific dental or oral surgery guidance exists. The aim of this paper is to review the current literature and guidance regarding the use of biologics in the perioperative period.
Key words: Biological products / infections / oral surgical procedures / oral surgery / postoperative complications
© The authors, 2023
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Biological therapies, also referred to as targeted immunomodulator therapies, are currently the fastest growing class of therapeutic products [1]. They have revolutionised the management of conditions such as rheumatoid arthritis, psoriasis, inflammatory bowel disease and oncological diseases, as their use has led to high rates of efficacy and remission. The growing use of biological agents in clinical practice has resulted in increasing numbers of these patients presenting in dental and maxillofacial settings [2].
Most synthetic drugs are small molecules with a precise chemical structure and made by a chemical process. Conversely, biological therapies are manufactured in a living system such as a microorganism, plant or animal complex [3]. They are protein-based molecules, or mixtures of molecules, often produced using recombinant DNA. Unlike corticosteroids, which are non-discriminant and suppress numerous host immune processes, biologics target specific elements of proinflammatory pathways [4]. Most are administered by injection, however the latest class of biologics, JAK inhibitors, can be taken orally. JAK inhibitors have a shorter half-life, so may offer increased flexibility and lower infection risk. Classes of biologics, their trade names and half-lives are outlined in Table I.
Biological agents do carry a risk of immunosuppression and have the potential to increase the risk of infections and delayed wound healing by blocking the normal inflammatory response in the perioperative period. However, stopping biologics may lead to flare up of the underlying condition which can affect postoperative recovery. There is little information regarding oral surgery procedures and biological therapy. Establishment of perioperative guidelines in oral surgery is needed as the number of patients on these medications increase.
Materials and methods
A literature search using Medline was completed using the MeSH terms ‘oral surgery’ and ‘biological products’. The search yielded 14 articles, none of which were relevant to management of biologics in the perioperative period. As there are currently no studies comparing interruption of biologics in oral and maxillofacial surgery, the search criteria was widened to assess perioperative management of biologics in all types of surgery. A search was performed using Medline and EMBASE databases with the following keywords: biologics, surgery, complications, postoperative outcomes, and perioperative interruption. This literature review was based on guidelines for the Preferred Reporting Items for systematic reviews and Meta-Analyses extension for Scoping Reviews (PRISMA-ScR) [10]. Articles were systematically screened to exclude irrelevant titles then full texts were assessed for eligibility. Subsequent results are presented in a PRISMA diagram (Fig. 1).
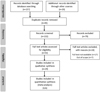 |
Fig. 1 PRISMA flow diagram demonstrating the screening and eligibility process during literature review. |
Results
There are currently no studies investigating the perioperative management of biological therapy for patients undergoing oral and maxillofacial surgical procedures. The majority of published evidence originates from retrospective studies of the rheumatoid arthritis population undergoing orthopaedic procedures. Of the 29 studies to date, 5 show statistically significant increased risk of infection when continuing biologics in the perioperative period, and 22 studies show no difference in postoperative outcomes (Tab. II).
The largest study to date was a retrospective study conducted by George et al. to assess postoperative infection following orthopaedic procedures in patients with rheumatology, dermatology and gastroenterology diseases [11]. The study compared patients who continued biological therapy in the perioperative period versus patients whose therapy was interrupted and found that administration of TNF-α inhibitor infliximab within four weeks of elective hip or knee arthroplasty was not associated with an increase in infection within 30 days.
One study investigated the impact of preoperative serum TNF-α inhibitor drug levels on postoperative outcomes in inflammatory bowel disease patients. It found that infectious complications were significantly higher in patients with therapeutic TNF-α inhibitor drug levels [12].
Most studies are retrospective, with heterogeneity of study methodologies, varying comparator groups and inconsistent outcome measures. These differences lead to variability in the association of infection with continued biological therapy in the perioperative period.
Currently no formal guidance on management of these patients perioperatively in dental or maxillofacial setting exists, but principles can be drawn from other specialties guidance. Several professional bodies have issued formal recommendations for management of patients receiving biological therapy in the perioperative period. The British Society for Rheumatology (2019) and the British Association of Dermatologists (2020) both agree on stopping biologic therapy for three to five times the half-life of the drug prior to major surgical procedures, and only restarting therapy postoperatively when there is no evidence of infection and wound healing is satisfactory [5,13]. The British Society for Rheumatology emphasise that the potential benefit of minimising postoperative infections should be balanced against a significant risk of flare in disease activity, as has been shown in some studies [39].
Discussion
By potentially inhibiting key molecules in normal inflammatory pathways, biologics may pose at least a theoretical risk of perioperative infection and delayed wound healing. The immune response plays an essential role during acute wound healing. The activation of immune cells and factors initiate the inflammatory process, facilitate wound cleansing and promote subsequent tissue healing [40]. Four overlapping phases are identified in acute wound healing; haemostasis, inflammation, proliferative phase and remodelling [41]. During the inflammatory phase TNF-α is expressed, and it includes stimulation of angiogenesis, fibroblast proliferation, and increasing collagenase and prostaglandin synthesis [42].
Perioperative interruption to biologics may reduce postoperative complication rates, but there is a need to balance the potential risk of infection with the risk of disease flare up and the potential that it may prove more difficult to re-establish disease control when a treatment has been temporarily withdrawn. Prediction of infectious or healing complications remains a challenge in these patients. There is an array of confounding factors which affect post-operative complications, including disease severity and flare up, comorbidities and immunosuppressive medications, and the type of surgery and the surgical stress response.
Disease severity and risk of flare up
The use of biologics is associated with high rates of improvement in disease signs and symptoms, but interruption to biological therapy can result in significant decline in disease control, which may require steroid therapy [5,42]. For patients with severe disease flare up, the consequences of this may outweigh the risk of post-operative infection, especially for minor procedures.
Comorbidities and immunosuppressive medications
Use of concurrent immunosuppressants (particularly corticosteroids and DMARDs), and the presence of other co-morbidities may contribute significantly to post-operative infection. Concurrent glucocorticoid use was associated with an increased risk of infection postoperatively [11]. Patients with comorbidities which cause immunosuppression, such as diabetes mellitus or cancer, will be at increased risk of complications and the management of their biological therapy requires careful consideration.
Surgery stimulates a stress response in the body, in which physiological neuro-endocrine and inflammatory-immune responses occur [43]. The degree of stress response will depend on the duration of surgery, its invasiveness and the extent of tissue injury. The greatest stress response is elicited from procedures such as total joint replacement, major abdominal, open vascular or cardiac surgery [44,45]. Minimally invasive surgical techniques, such as laparoscopic surgeries, reduce the inflammatory response and associated tissue injury. Stress response is one means of quantifying the relative risk of a procedure, within which many oral and maxillofacial surgeries could be considered lower risk.
Recommendations
Management of biologics should be an individualised decision, taking into account disease activity, risk of flare up, comorbidities, concurrent immunosuppressive therapy, the type of surgery and patient preference. Following patient assessment, elective procedures can be either scheduled at the end of the dosing interval where the immunosuppressive effects of the drugs would be at their lowest, or biological therapy can be interrupted for three to five times the half-life before surgery based on dosing interval and half-life of the individual drug. When making this decision, it is advisable to liaise with the patient's prescribing physician and the patient on the best course of action.
When biologic therapy is interrupted, it can be restarted postoperatively if wound healing is satisfactory, sutures and staples are removed, and there are no clinical signs of infection. An outpatient review should be arranged approximately fourteen days following surgery for a clinician to assess healing. With regards to emergency surgery, scheduling and therapy interruption are not possible, so patients should be closely monitored for infection or other complications postoperatively.
Conclusion
Biologics are increasingly being used for management of immune-mediated inflammatory disorders. Management of these agents around the time of surgery requires balancing the risk of infection and delayed wound healing against the risk of disease flare up. Guidelines have provided a starting point to minimise perioperative risk, but recommendations should be considered in the context of the individual. Good communication between the surgical team and the prescribing physician is required to ensure management of biologics in the perioperative period carries optimal outcomes for patients. Further research is required to determine the implications of biologics on postoperative infection in oral surgery and a position paper is necessary to shape professional guidance.
Conflict of interest
The authors declare that they have no conflicts of interest in relation to this article.
Funding
The authors declare having received no funding for this article.
Ethical approval
Ethical approval will not be required because this study retrieved and synthesised data from previously published studies.
Informed consent
As this study systematically reviewed previously published studies, no new informed consent was required but ethical principles were followed.
Authors contributions
Sophie Mills, Patrick Ryan and Karl Gaffney contributed equally to the manuscript, with Sophie Mills writing the first draft and all authors participating in the editing process and approving the final version.
References
- Food US, Administration D. Commissioner Scott Gottlieb. The FDA's Biosimilars Action Plan 2018. Brookings Institution, Washington, DC. https://www.fda.gov/news-events/press-announcements/remarks-fda-commissioner-scott-gottlieb-md-prepared-delivery-brookings-institution-release-fdas [Accessibility verified November 14, 2021] [Google Scholar]
- O'Neill ID, Scully C. Biologics in oral medicine: principles of use and practical considerations. Oral Dis 2012;18:525–536. [CrossRef] [PubMed] [Google Scholar]
- Ho DY, Subramanian AK. Infections related to biologics. Infect Dis Clin N Am 2020;34:xiii–xvi. [Google Scholar]
- Zaghiyan K, McGovern D, Fleshner P. Should biologic agents be stopped before surgery for inflammatory bowel disease? Expert Rev Gastroenterol Hepatol 2015;9:269–272. [CrossRef] [PubMed] [Google Scholar]
- Smith CH, Yiu ZZN, Bale T, et al. British Association of Dermatologists guidelines for biologic therapy for psoriasis 2020-a rapid update. Br J Dermatol 2020;183:628–637. [CrossRef] [PubMed] [Google Scholar]
- Goodman SM, Springer B, Guyatt G, et al. American College of Rheumatology/American Association of Hip and Knee Surgeons guideline for the perioperative management of antirheumatic medication in patients with rheumatic diseases undergoing elective total hip or total knee arthroplasty. Arthritis Rheumatol 2017;69:1538–1551. [CrossRef] [PubMed] [Google Scholar]
- Lortholary O, Fernandez-Ruiz M, Baddley JW, et al. Infectious complications of rheumatoid arthritis and psoriatic arthritis during targeted and biological therapies: a viewpoint in 2020. Ann Rheum Dis 2020;79:1532–1543. [CrossRef] [PubMed] [Google Scholar]
- Abou Zahr Z, Spiegelman A, Cantu M, et al. Perioperative use of anti-rheumatic agents does not increase early postoperative infection risks: a Veteran Affairs' administrative database study. Rheumatol Int 2015;35:265–272. [CrossRef] [PubMed] [Google Scholar]
- Bafford AC, Powers S, Ha C, et al. Immunosuppressive therapy does not increase operative morbidity in patients with Crohn's disease. J Clin Gastroenterol 2013;47:491–495. [CrossRef] [PubMed] [Google Scholar]
- Scott DL. Biologics-based therapy for the treatment of rheumatoid arthritis. Clin Pharmacol Ther 2011;91:30–43. [Google Scholar]
- George MD, Baker JF, Hsu JY, et al. Perioperative timing of infliximab and the risk of serious infection after elective hip and knee arthroplasty. Arthritis Care Res (Hoboken) 2017;69:1845-1854. [CrossRef] [PubMed] [Google Scholar]
- Lau C, Dubinsky M, Melmed G, et al. The impact of preoperative serum anti-TNFα therapy levels on early postoperative outcomes in inflammatory bowel disease surgery. Ann Surg 2015;261:487-496. [CrossRef] [PubMed] [Google Scholar]
- Holroyd CR, Seth R, Bukhari M, et al. The British Society for Rheumatology biologic DMARD safety guidelines in inflammatory arthritis. Rheumatology 2019;58:e3–e42. [CrossRef] [PubMed] [Google Scholar]
- Bibbo C, Goldberg JW. Infectious and healing complications after elective orthopaedic foot and ankle surgery during tumor necrosis factor-alpha inhibition therapy. Foot Ankle Int 2004;25:331–335. [CrossRef] [PubMed] [Google Scholar]
- Bongartz T, Halligan CS, Osmon DR, et al. Incidence and risk factors of prosthetic joint infection after total hip or knee replacement in patients with rheumatoid arthritis. Arthritis Rheum 2008;59:1713–1720. [CrossRef] [PubMed] [Google Scholar]
- den Broeder AA, Creemers MC, Fransen J, et al. Risk factors for surgical site infections and other complications in elective surgery in patients with rheumatoid arthritis with special attention for anti-tumor necrosis factor: a large retrospective study. J Rheumatol 2007;34:689–695. [PubMed] [Google Scholar]
- Cohen BL, Fleshnet P, Kane SV, et al. Anti-tumor necrosis factor therapy is not associated with post-operative infection: results from prospective cohort of ulcerative colitis and Crohn's disease patients undergoing surgery to identify risk factors for postoperative infection. Gastroenterology 2019;156:S80. [Google Scholar]
- Gainsbury ML, Chu DI, Howard LA, et al. Preoperative infliximab is not associated with an increased risk of short-term postoperative complications after restorative proctocolectomy and ileal pouch-anal anastomosis. J Gastrointest Surg 2011;15:397–403. [CrossRef] [PubMed] [Google Scholar]
- George MD, Baker JF, Winthrop K, et al. Timing of abatacept before elective arthroplasty and risk of postoperative outcomes. Arthritis Care Res 2019;71:1224–1233. [CrossRef] [PubMed] [Google Scholar]
- Giles JT, Bartlett SJ, Gelber AC, et al. Tumor necrosis factor inhibitor therapy and risk of serious postoperative orthopaedic infection in rheumatoid arthritis. Arthritis Rheum 2006;55:333–337. [CrossRef] [PubMed] [Google Scholar]
- Godot S, Gottenberg JE, Paternotte S, et al. Safety of surgery after rituximab therapy in 133 patients with rheumatoid arthritis: data from the autoimmunity and rituximab registry. Arthritis Care Res (Hoboken) 2013;65:1874–1879. [CrossRef] [Google Scholar]
- Hirao M, Hashimoto J, Tsuboi H, et al. Laboratory and febrile features after joint surgery in patients with rheumatoid arthritis treated with tocilizumab. Ann Rheum Dis 2009;68:654–657. [CrossRef] [PubMed] [Google Scholar]
- Hirano Y, Kojima T, Kanayama Y, et al. Influences of anti-tumour necrosis factor agents on postoperative recovery in patients with rheumatoid arthritis. Clin Rheumatol 2010;29:495–500. [CrossRef] [PubMed] [Google Scholar]
- Holubar S, Eisenstein S, Bordeianou L, et al. Biologics before surgery for IBD: are they associated with post-operative infectious complications? Results from the national surgical quality improvement program inflammatory bowel disease collaborative in >1500 patients. Inflamm Bowel Dis 2021;27:S57– S58. [Google Scholar]
- Kawakami K, Ikari K, Kawamura K, et al. Complications and features after joint surgery in rheumatoid arthritis patients treated with tumour necrosis factor-α blockers: perioperative interruption of tumour necrosis factor-a blockers decreases complications? Rheumatology 2010;49:341–317. [CrossRef] [PubMed] [Google Scholar]
- Kim JY, Zaghiyan K, Fleshner P. Risk of postoperative complications among ulcerative colitis patients treated preoperatively with vedolizumab: a matched case-control study. BMC Surg 2020;20:46. [CrossRef] [PubMed] [Google Scholar]
- Kubota A, Nakamura T, Miyazaki Y, et al. Perioperative complications in elective surgery in patients with rheumatoid arthritis treated with biologics. Mod Rheumatol 2012;22:844–848. [CrossRef] [PubMed] [Google Scholar]
- Kunitake H, Hodin R, Shellito PC, et al. Perioperative treatment with infliximab in patients with Crohn's disease and ulcerative colitis is not associated with an increased rate of postoperative complications. J Gastrointest Surg 2008;12:1730–1737. [CrossRef] [PubMed] [Google Scholar]
- Kulaylat AN, Kulaylat AS, Schaefer EW, et al. The impact of preoperative anti-tnfα therapy on postoperative outcomes following ileocolectomy in Crohn's disease. J Gastrointest Surg 2021;25:467–474. [CrossRef] [PubMed] [Google Scholar]
- Latourte A, Gottenberg JE, Luxembourger C, et al. Safety of surgery in patients with rheumatoid arthritis treated by abatacept: data from the French Orencia in Rheumatoid Arthritis Registry. Rheumatology (Oxford) 2017;56:629–637. [Google Scholar]
- Marchal L, D'Haens G, van Assche G, et al. The risk of post-operative complications associated with infliximab therapy for Crohn's disease: a controlled cohort study. Aliment Pharmacol Ther 2004;19:749–754. [CrossRef] [PubMed] [Google Scholar]
- Mitsuya JB, Gonzalez R, Thomas R, et al. The effect of biologics on postoperative complications in children with inflammatory bowel disease and bowel resection. J Pediatr Gastroenterol Nutr 2019;68:334–338. [CrossRef] [PubMed] [Google Scholar]
- Momohara S, Kawakami K, Iwamoto T, et al. Prosthetic joint infection after total hip or knee arthroplasty in rheumatoid arthritis patients treated with non-biologic and biologic disease-modifying antirheumatic drugs. Mod Rheumatol 2011;21:469–475. [CrossRef] [PubMed] [Google Scholar]
- Ruyssen-Witrand A, Gossec L, Salliot C, et al. Complication rates of 127 surgical procedures performed in rheumatic patients receiving tumor necrosis factor alpha blockers. Clin Exp Rheumatol 2007;25:430–436. [PubMed] [Google Scholar]
- Talwalkar SC, Grennan DM, Gray J. Tumour necrosis factor antagonists and early postoperative complications in patients with inflammatory joint disease undergoing elective orthopaedic surgery. Ann Rheum Dis 2005;64:650–651. [CrossRef] [PubMed] [Google Scholar]
- Waterman M, Xu W, Dinani A, et al. Preoperative biological therapy and short-term outcomes of abdominal surgery in patients with inflammatory bowel disease. Gut 2013;62:387–394. [CrossRef] [PubMed] [Google Scholar]
- Wendling, D. Surgery in rheumatoid arthritis patients under anti-TNF-α therapy: what is the risk? Clin Rheumatol 2007;26:1396–1397. [CrossRef] [PubMed] [Google Scholar]
- Bakkour W, Purssell H, Chinoy H, et al. The risk of post-operative complications in psoriasis and psoriatic arthritis patients on biologic therapy undergoing surgical procedures. J Eur Acad Dermatol Venereol 2016;30:86–91. [CrossRef] [PubMed] [Google Scholar]
- Raziyeva K, Kim Y, Zharkinbekov Z, et al. Immunology of acute and chronic wound healing. Biomolecules 2021;11:700. [CrossRef] [PubMed] [Google Scholar]
- Larouche J, Sheoran S, Maruyama K, et al. Immune regulation of skin wound healing: mechanisms and novel therapeutic targets. Adv Wound Care (New Rochelle) 2018;7:209–231. [CrossRef] [PubMed] [Google Scholar]
- Anstead GM. Steroids, retinoids, and wound healing. Adv Wound Care 1998;11:277–285. [PubMed] [Google Scholar]
- Au K, Reed G, Curtis JR, et al. High disease activity is associated with an increased risk of infection in patients with rheumatoid arthritis. Ann Rheum Dis 2011;70:785–791. [CrossRef] [PubMed] [Google Scholar]
- Cusack B, Buggy DJ. Anaesthesia, analgesia, and the surgical stress response. BJA Educ 2020;20:321–328. [CrossRef] [PubMed] [Google Scholar]
- Watt DG, Horgan PG, McMillan DC. Routine clinical markers of the magnitude of the systemic inflammatory response after elective operation: a systematic review. Surgery 2015;157:362–380. [CrossRef] [PubMed] [Google Scholar]
- McInnes IB, Schett G. The pathogenesis of rheumatoid arthritis. N Engl J Med 2011;365:2205–2219. [Google Scholar]
All Tables
All Figures
 |
Fig. 1 PRISMA flow diagram demonstrating the screening and eligibility process during literature review. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


