| Issue |
J Oral Med Oral Surg
Volume 28, Number 1, 2022
|
|
|---|---|---|
| Article Number | 14 | |
| Number of page(s) | 9 | |
| DOI | https://doi.org/10.1051/mbcb/2021038 | |
| Published online | 08 March 2022 | |
Original Research Article
Intra-socket application of Hyaluronic acid reduces pain and swelling following wisdom teeth removal
1
Oral and Maxillofacial Surgery, Department of Oral & Maxillofacial Surgery, Faculty of Dentistry, Mahidol University, 6 Yothi Street, Rachathewee District, Bangkok 10400, Thailand
2
Walailak University International College of Dentistry, 979/42–46 Phahonyothin Road, 19th floor SM Tower, Phaya Thai District, Bangkok 10400, Thailand
3
Private practice
4
International College of Dentistry Committee, Walailuk University International College of Dentistry, 979/42–46 Phahonyothin Road, 19th floor SM Tower, Phayathai District Bangkok 10400, Thailand
* Correspondence: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
18
August
2020
Accepted:
1
September
2021
Abstract
Background: Hyaluronic acid (HA) has the potential to promote wound healing. Pain and swelling with trismus are common sequalae post wisdom teeth removal. This study aims to investigate the effectiveness of intra-socket HA solution to reduce these uncomfortable post-operative events. Materials and methods: 30 patients underwent bilateral extractions of mandibular wisdom teeth for this study. Intra-socket application of 0.7 ml 20 mg/2 ml HA solution (Hyalgun) with Gel foam as a scaffold in study site versus Gel foam only on control site was conducted via a split mouth study design. Data collection of five facial reference points for swelling and maximum mouth opening was recorded during the pre-operative period and post-operative 2nd and 7th day. The VAS pain score at post-op 1st, 2nd and 3rd day and the number of analgesics for the 7-day post-operative duration were evaluated. Results: The HA group demonstrated statistically significant less swelling, trismus and analgesia consumption on the 2 and 7 days after surgery. VAS scores on day 1, 2 and 3 after surgery (P = 0.05) were significantly less in the HA group compared to the control group. Conclusion: The application of intra-socket HA has a positive effect for reducing postoperative pain and swelling with trismus after the lower third molar intervention (LTMI).
Key words: Hyaluronic acid / lower 3rd molar surgery / post-operative pain / swelling / trismus
© The authors, 2022
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Background and Rationale
Mandibular third molar removal is one of the most widely done treatments in the oral cavity. Nevertheless, surgeons often encounter undesirable post-operative sequalae. These inflammatory responses after wisdom teeth surgery may badly affect a patients' quality of life [1–3]. Post-operative complications like dry socket, nerve damage, bone fracture, delayed healing and damage of the second molar can also happen quite frequently following the surgical removal of mandibular third molars [4,5].
The lower third molar surgery usually entails the elevation of a full thickness gingival flap and guttering of bone surrounding the tooth, this causes a significant post-operative downtime which has been documented to reduce the patients' quality of life [2,6]. In 1997 Berge [7] published a study in Norway where 57% of patients who had underwent wisdom teeth removal could not turn up for work for an average of 1.07 days.
Numerous surgical techniques and materials that focused on decreasing complications and accelerating healing after (lower third molar intervention) LTMI have been well documented [8–17]. Local/ systemic steroids, Non-steroidal Anti-inflammatory drugs, and antibiotics are medicinal options that are frequently used and seem to be effective in increasing postoperative quality of life after impacted LTMI [18]. Nonetheless, the routine prescription of these drugs can cause undue problems because of their potential adverse side effects. Furthermore, these medications could be contraindicated for some patients.
The corticosteroids help to decrease pain, edema and trismus from LTMI; and short period usage of corticosteroid may usually show no adverse effect; but many practitioners do not prefer it as it may delay wound healing [1–3]. These all issues stimulated researchers to search for other options with less or no systemic side effects to reduce post-operative sequelae in the post-surgery socket. Nowadays, the expectations of patients have dramatically increased; common sequelae and complications such as swelling and trismus that were once taken for granted may be met with harsh reviews and sometimes even unwarranted legal claims. Therefore, to build a successful practice or oral surgery department, one should always be on the lookout for better medications or techniques which have the potential to expedite and improve the healing process.
The advent of regenerative bio-medicines in the 21st century has shown promising results in treating patients with debilitating tissue damage, diseases and aging [19]. The field of regenerative medicine has discovered the effectiveness of some biomaterials in mimicking the complex environment of native tissues to restore, maintain, or improve tissue function as well as wound healing and tissue engineering [20].
Hyaluronic acid (HA) is a versatile naturally available material that plays a multifaceted role in biology (10) It has shown outstanding results in accelerating recovery by means of inducing fast granulation tissue production, restricting the destruction from inflammation, and inducing re-epithelialization with angiogenesis [21,22]. HA also possesses non-immunogenic and non-toxic characteristics, thus, making it a safe material to serve in many medical applications [10].
In 1997 Pagnacco and Vangelisti conducted a clinical trial of HA in dentistry for the treatment of periodontal disease [23]. It is a not only a key element in periodontal tissues such as gingiva, and periodontal ligament but also hard tissues like alveolar bone and cementum [24], therefore, it should be an ideal product for intraoral wound recovery.
The HA solution used in the treatment of temporomandibular joint diseases follow arthrocentesis found its place as the first application in oral and maxillofacial surgery. From then still now it used widely in TMJ disorder as it is the physiological compound in synovial fluid which lubricates and decreases articular wear. The metabolic properties of HA favor revascularization to the TMJ anatomical structures, therefore, it is effective in treating various TMJ disorders by improving joint function and decreasing the pain [22,25].
Many previous studies including Nadia et al. [26] have reported about the effectiveness of HA in the treatment of gingivitis, recessions, periodontal pockets, grafts and implants [27,28]. To date, the use of HA has no known deleterious side effects nor adverse reactions with other drugs [29,30].
Many previous studies regarding the efficiency of HA in different forms like spray, gel have shown promising results in terms of reduction of post-operative sequelae after LTMI [10,26,31–32].
To date, all previous studies relating to HA for TMJ arthrocentesis used 0.5 ml or 0.75 ml or 1ml with 20 mg/2 ml or 10 mg/1 ml solution and no side effects from HA were found among these studies [33–37].
Corticosteroids and HA have both been used to improve pain, trismus and masticatory function post LTMI and TMJ disorders. But it has been postulated that the use of HA is superior to corticosteroid because it is a physiological component without risk of degenerative properties as it would with corticosteroids [38].
In this research, our objective was to investigate the effectiveness of HA for reducing pain, limited mouth opening and edema following the removal of impacted mandibular third molars.
Materials and methods
Study design
Our study was done from December 2018 to December 2019. A prospective, randomized, split-mouth, double-blind cross over clinical trial with 30 patients were recruited in the Department of Oral and Maxillofacial Surgery clinic, Faculty of Dentistry, Mahidol University for this study. Figure 1. The study comprised with the patient having bilateral impacted LTM who needs surgical extraction. The degrees of surgical difficulty calculated for the LTMI based on LTM Pederson scale [39].
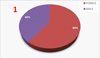 |
Fig. 1 Male-Female ratio in this study who assessed pre and post-operatively all 3 clinical parameters. |
Ethical approval
The study followed the Declaration of Helsinki. The Ethics committee approved our study, (Mahidol University Faculty of Dentistry Ethics Committee, protocol code COA.NO.MU-DT/PY-IRB 2019/043.0507).
Sampling technique
Sample size calculation performed by using G power 3.1.0 software, assuming α error = 0.05, power = 95% and estimated effect size = 0.4, after our pilot study we calculated our sample size following the related formula recommendations. Final sample size 30 adult volunteers. along considering with approximate 20% dropouts. All of the patients had bilaterally impacted lower third molars. The inclusion criteria contain the patients has impacted third molars symmetrically positioned on both sides of the mandible, Age between 18 to 40 years, patients classified as physical status-I using the guide line of American society of Anesthesiologist, non-smoker and not alcoholic, patients included must have upon presentation bilateral impacted lower third molars, which require flap opening, bone removal and tooth separation during the operation . The exclusion criteria were with the patients having systolic blood pressure (>140 mmHg, <90 mmHg) and diastolic blood pressure (>90 mmHg, <60 mmHg), to use contraceptives or corticosteroids which can affect the postsurgical healing phase and amount of swelling on the face, to have difficulty with co-operation, Inability to follow the instructions during the study, pregnancy or current lactation, to have acute infection such as pericoronitis and/or pain on the tooth site before extraction to take antibiotics 15 days before surgery or any medication during the previous 5 days prior to the surgery that would alter their perception of pain (analgesic, antidepressants), allergic to local anesthetics, avian protein, Feathers, Egg products and the facial deformities that may interfere with the injections, surgery or evaluation. The participants were randomly divided into 2 groups by random table method. Each patient was randomly assigned two different post-extraction intra-socket products which we used HA + gelfoam in study group and gelfoam alone in control group. After surgical removal of one-sided lower 3rd molar (either right or left side), we waited 3–4 week for the removal of another side for this split mouth study. The patient had their full freedom to withdrawal their participation in the study at any time depending on their own decision.
Methods
Pre-surgical assessments
The day of operation patient arrived oral and maxillofacial surgery department about 1 hour before the operation start. Base line swelling and trismus measured. Patient was explained for understanding the VAS clearly. The degrees of surgical difficulty calculated before the extractions by a single investigator. The degrees of surgical difficulty calculated for the LTMI before extraction based on LTM Pederson scale [39]. The surgical extractions according to difficulty were classified as easy, moderate, or difficult (Tab. I).
Criteria and scores of the Pederson scale.
Surgical procedure
The same oral and maxillofacial surgeon performed the surgical procedures. Patients and examiners were blinded to the allocation of the HA throughout the study. At first local anesthesia through inferior alveolar nerve block, lingual nerve block, and long buccal nerve infiltration were performed using 4% articaine with 1:1,00,000 epinephrine. A full thickness Envelope Flap was done for all 30 wisdom teeth, the bone was guttered with saline irrigation and tooth was split to eliminate obstructions in the path of removal.
Application of injected form of HA
After the surgical extraction, copious irrigation of the extraction socket was done, another coauthor then applied 0.7 ml from 20 mg/2 ml Hyalgun into the gelfoam and gently placed it intra-socket. On the control side, only gelfoam was placed intra-socket with primary wound closure (Fig. 2). Only when either Gelfoam alone or HA plus Gelfoam was blood-soaked, was the surgeon called back for suturing, this is to ensure the operator is blinded from knowing which material was used. Hyalgun is commercial form of HA solution from the Healthcare Pharmaceuticals company of Bangladesh. It contains Sodium Hyaluronate in a viscous solution that contain a high molecular weight fraction of purified sodium hyaluronate (HA) in buffered physiological sodium phosphate, each pre-filled syringe has Sodium Hyaluronate BP 20 mg/2 ml as solution. As well as gelfoam is also commercially available form from Thailand called SURGISPON. We used gelfoam in both study and control group as following previous study [32], as it acts as a scaffold to hold the HA in socket and also as a placebo in control group.
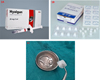 |
Fig. 2 (A) Hyalgun (Hyaluronic acid solution 20 mg/2 ml), (B) Gelfoam, (C) Hyaluronic acid solution applied with gelfoam. |
Postoperative management
After the LTIM, they were given a carry-home copy of the case chart. Instructions for post-surgical care were given in written and verbal forms. All the patients were prescribed amoxicillin 500 mg orally 4 times a day for 5 days and if allergic to penicillin then clindamycin 300 mg 4 times a day for 5 days. Paracetamol 500 mg in case of pain as a main analgesia and also prescribed tramadol 50 mg orally as only for rescue analgesia.
Patients were instructed to record the amount and time the paracetamol was taken at home until the end of first postoperative week. Additionally, the investigator called patients by telephone to remind them to follow the instructions that had been given to them.
Clinical parameters
A. Evaluation of swelling
The facial swelling measurements were determined based on the previous article of Latt et al. (Fig. 3) for both study and control group [39]. The following measurements were made between five landmarks:
Gonion to lateral canthus of eye.
Tragus to angle of mouth.
Tragus to pogonion.
The average of the three measurements were calculated. Measurements were done immediately before surgery and on the 2nd and 7th post-operative day.
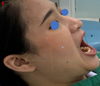 |
Fig. 3 Facial swelling and trismus measurements. A: Gonion to lateral canthus of eye; B: Tragus to angle of mouth; C: Tragus to pogonion; D: Maximum inter-incisal. |
B. Evaluation of trismus
The trismus evaluation was predictable by measuring maximum inter-incisal distance (distance between upper and lower incisal edge of central incisors) using Vernier caliper. We calculated trismus for both study and control side by measuring from the edges of the upper central incisor to the lower right central incisors during maximum opening of the patients with Vernier calipers at the immediate preoperative period and on the 2nd and 7th post-operative day (Fig. 3).
C. Evaluation of post-surgical pain
The VAS pain score was used in this study. Participants recorded their pain and we changed NRS instead of VAS pain score as a number “pain-free” (0–4 mm), “mild pain” (5–44 mm), “moderate pain” (45–74 mm), and “severe pain” (75–100mm). Moreover, it contains facial expression illustrations to help direct and instruct the patients. In our study, postoperative pain measurements were recorded by using pain score in the immediate postoperative period, the first, second and third post-operative days, whilst there are no pain measurements taken from post-operative day 4–7.
In addition, patients recorded the amount of paracetamol taken daily after the LTMI. We prescribed paracetamol 500 mg as a pain medicine, we instructed all patients to take paracetamol for pain control and to only ingest tramadol 50 mg if pain was unbearable even after paracetamol intake. The daily total paracetamol intake during the 7days following LTMI was recorded.
The examiner is the same person who assessed pre and post-operatively all 3 clinical parameters.
Statistical analysis
The statistical analysis was performed using the PASW Statistics 18.0 (SPSS Inc. Chicago.IL, USA). The Descriptive statistics including mean values and standard deviations calculated for all variables in the study and control groups. Repeated measured ANOVA used for measurement of trismus and swelling. Wilcoxon test was used for non-parametric distributed variables (VAS scores). Paired T-test was used to compare the statistical differences of number of analgesic tablets taken between HA group and control group. P-value of less than 0.05 was accepted as significant.
Results
The 30 patients among them 18 females and twelve males completed this study. The mean age was 22 years with the age range: 18–40 years.
Mean surgical duration
The duration of surgery was recorded from the time of first incision to insertion of final suture. The mean duration was 31.09 ± 3.987 min for HA group and 30.60 ± 2.859 min for the control group.
Facial swelling measurement (Tab. II)
Pre-operatively the baseline measurements in the study group were (12.37 ±.66 mm) and control group was (12.43 ± 0.70 mm) and there was no statistically significant difference between both groups with P value 0.388.
On the second postoperative day, it increased significantly in both groups compared to preoperative measurements; but in the HA group facial swelling was comparatively lower (12.76 ±.32) than the control group (13.95 ±.65). The difference between the two groups was statistically significant with P value is 0.001
By the seventh postoperative day, facial swelling reduced in both groups, in the study group swelling was (12.47 ±.46) and control group (12.76 ±.68 mm) which also showed a statistically significant difference between the 2 groups with P value is 0.016.
Evaluation of Swelling in this study as mean ± standard deviation.
Maximum mouth opening measurement (Tab. III)
Maximal inter-incisal distance levels were found almost similar preoperatively for both two groups.
Immediate pre-operative measurement of trismus value is (37.55 ± 4.98 mm) and (37.77 ± 5.67 mm) for study and control group respectively with P-value 0.798 which is not statistically significant.
Although both groups developed trismus within the 2nd post-operative day, our results found statistically significant higher mouth opening in the study group (26.07 ± 4.42) compared to the control group (21.62 ± 5.50) with P value 0.001.
However, the mouth opening increases within the end of the week for both groups, but here also significant difference was found between HA and control group. On the seventh postoperative day, the mouth opening in HA Group was (34.24 ± 5.14 mm) and control group was (29.10 ± 5.92 mm) with P value 0.001 which showed statistical significance.
Evaluation of trismus as mean ± standard deviation (median) in mm.
Pain measurement (Tabs. IV and V)
In regard to the mean VAS scores, there was a significant difference between the HA and control group for pain on the first, second, and third days after LTMI, according to the 100 mm VAS (Tab. IV) with P value is 0.001, 0.002, 0.002 on Day 1, 2 and 3 respectively.
Total analgesic consumption also noted for the first 7 days post LTMI. We prescribed standing doses of paracetamol 500 mg for reducing pain and tramadol 50 mg prescribed only for rescue purpose, in this study we found only 2 patients that took tramadol 50 mg on the control side of the split-mouth study, we calculated overall analgesic consumption only for Paracetamol but not for tramadol. The results show that the HA group took significantly lesser analgesics compared to the control group with P value is 0.001 (Tab. V).
Evaluation of pain by VAS score with 30 the patients.
Total numbers of analgesic consumption in 30 patients for analgesics in 3 days.
Discussion
Wisdom teeth removal especially in deep impacted cases is often marred by severe postoperative inflammatory reactions such as pain, swelling and trismus. It is in our best interest to find ways to mitigate these sequalae following LTMI.
Several previous studies [40,41] reported that post-operative sequalae vary depending on the age, gender and surgical difficulty of the impacted teeth. All of these variables were equally matched based on a split mouth study design on 30 patients with symmetrical bilateral mandibular wisdom teeth. Post LTMI intra-socket application of 0.7 ml from 20 mg/2 ml HA solution (Hyalgun) with gelfoam in study group and only intra-socket gelfoam in the control group after LTMI was done. As this is the first ever study regarding intra-socket Hyalgun application in LTMI, there is no consensus regarding the appropriate dose and protocol of HA, since to date no comparison has been made among them. All of the previous studies used different doses such as 0.5 ml or 0.7 ml or 1 ml from 20 mg/2 ml or 10 mg/1ml Hyalgun with no side effects [33–37]. Among them some studies found that 0.7ml Hyalgun showed better outcome when compared to a control group [41,42] regarding pain and other associated symptoms. Therefore, in the same vein, we used 0.7 ml from 20 mg/2 ml Hyalgun.
In 2014 the previous study of Koray et al. [10] and in 2016 the article of Merchant [43] showed that HA spray can significantly reduce swelling and trismus compared to control group, but no role in controlling pain. This could be because the ‘spray’ form could only act on the superficial surface of the mucosa which does not offer enough contact with an extraction socket.
In 2016 another study of Yilmiz et al. [31] regarding HA on 3rd molar extraction sites showed beneficial effect in terms of reducing pain but not on swelling and trismus. In our study all post-op parameters showed significantly less swelling, pain and trismus in the experiment group compared to the control group. This could be attributed to the higher concentration of HA in our study (0.7 ml) as opposed to the (0.5 ml) which was used by Yilmiz et al.
This study evaluated facial swelling [44] measurements pre-operatively, post LTIM day 2, and day 7. The results showed a significant difference in the swelling levels between the study and control group (Tab. II). The swelling in the control group was comparatively more than the study group on postoperative day 2, and day 7. This study also found that the mouth opening was significantly higher in study Group than in control group on post-operative day 2, indicating less trismus in the study group. Although the mouth opening improved within the end of the week for both groups, the improvement was more significant in the study group compared to control group.
Koray et al. [10] and Merchant in 2016 [43] both of their studies evaluated the efficiency of HA in on post-operative pain, swelling and trismus and both of the studies showed that mouth opening was significantly greater and swelling is significantly less in HA group when compared to control group.
Afat [45] evaluated the effectiveness of leukocyte platelet-rich fibrin (L-PRF) versus (L-PRF) combined with a HA sponge on sequalae after LTMI. The results showed that L-PRF in combination with HA, has the potential to reduce swelling after LTMI.
In 2018 Bayoum et al. [31] documented that HA provides a positive impact on postoperative swelling and trismus after LTMI.
For all these beneficial reasons the use of HA has been recognized in various sites and conditions in the oral cavity. In 2008 the study of Lee [46] showed that HA gel on oral ulcers helped reduce overall signs and symptoms. HA also accelerated the healing period and decreased pain for both bechet disease and aphthous ulcer.
Romeo et al. [47] reported that HA gel together with amino acids improved secondary wound closures in patients undergoing excisional oral biopsy. Gontiya [27] documented that cross-linked HA along with SRP reduced gingival bleeding indices in chronic periodontitis patients.
Chang et al. [48] reported HA improved bone healing in an in-vitro study in a calvarian bone defect model.
Our study found remarkably lesser pain in the study group versus control group both in terms of VAS and analgesic consumption rate. The pain score reached its highest peak within the 2nd post-operative day, the pain score based on the VAS scale in the study group showed significantly lower scores than the control group P value < .005 Within 7 post-operative days, pain gradually decreased, for both groups with lesser pain in the study group.
Tramadol 50 mg was taken by only 2 patients after LTIM on the control side (intrasocket Gelfoam) procedure. Sockets in the study group did not require tramadol for pain management, this could be ascribed to the HA, which decreases pain by accelerating the healing process.
Grond and Sablotzki [49] mentioned that 50 mg tramadol acts a painkiller without antiedema properties during the post-operative period after impacted LTMI. With this in mind, the administration of tramadol did not interfere with our outcome variables regarding swelling and trismus. This current study showed that the mean pain VAS scores peaked on the day following the procedure but they were significantly lower in the study group by the 7th postoperative day.
Some post-operative complications like alveolar osteitis and postoperative infections may occur after LTMI with the ranges from 25–30% and 2–12% respectively. This study also prescribed antibiotics for both groups to prevent this complication. In this study there were no postoperative complications after 60 LTMI.
Although, this current study found beneficial effects by the use of HA solution to reduce pain, swelling and trismus after impacted LTMI, there were also several limitations:
We had a small sample size.
Although the study showed that the application of HA solution provided positive effects on VAS pain scale and an overall reduction in daily and total use of analgesics consumption, the evaluation of pain with VAS scores relied on the subjective evaluation of the patient's pain perception, which could be highly influenced by the patient's pain threshold, current state of mind, and past experiences.
There are a limited number of clinical studies with HA in the oral region and to the best of this authors knowledge this is the very first study about Hyalgun in LTMI, further randomized trials should be designed with more participants to evaluate the efficacy of HA.
Conclusion
Our study suggests that intra-socket HA solution is a promising approach to minimize postoperative pain and trismus from edema after LTMI, thus, it could be an additional option for oral surgeons to use for the patient's post-operative comfort. Additionally, Intra-socket HA reduced the usage of painkillers after LTMI. Although our study shows promising results with intra-socket HA post LTMI, further research with a larger sample size is imperative for drawing finite conclusions.
Declaration of patient consent
The authors certify that they have obtained all appropriate patient consent forms. In the form, the patient(s) has/have given his/her/their consent for his/her/their images and other clinical information to be reported in the journal. The patients understand that their names and initials will not be published and due efforts will be made to conceal their identity, but anonymity cannot be guaranteed.
Clinical Trial Register
No Requirement in our faculty because the clinical research is controlled by the Committee in the Ethics of Research in Human Being of Dentistry and Pharmacy Mahidol University Institutional Review Board.
Funding
This research received no external funding or any scholarship.
Conflicts of interest
The authors declare no conflict of interest.
Ethic approval
The study followed the Declaration of Helsinki in terms of medical protocol and ethics. Ethics committee approval for this clinical investigation obtained from the appropriate institution (Mahidol University Faculty of Dentistry Ethics Committee, protocol code COA.NO.MU-DT/PY-IRB 2019/043.0507).
Author Contributions
Conceptualization: Teeranut Chaiyasamut, Lee Kian Khoo, Nadia Sultana Shuborna; methodology: Nadia Sultana Shuborna; validation: Bishwa Prakash Bhattarai; formal analysis: Nadia Sultana Shuborna; investigation: Bishwa Prakash Bhattarai, Lee Kian Khoo; resources: Nadia Sultana Shuborna; data curation: Nadia Sultana Shuborna; writing—original draft preparation: Bishwa Prakash Bhattarai; writing—review and editing: Natthamet Wongsirichat, Teeranut Chaiyasamut; visualization: Verasak Pairuchvej, Teeranut Chaiyasamut, Lee Kian Khoo; supervision,: Sirichai Kiattavorncharoen, Natthamet Wongsirichat, Verasak Pairuchvej; project administration: Sirichai Kiattavorncharoen, Natthamet Wongsirichat, Teeranut Chaiyasamut.
Acknowledgments
Firstly, I would like to express my deep respect to my major advisor, for his valuable advice and kindness in guiding me during my research work. I will be forever thankful for his mentorship, constant support, and helpful counsel. My special gratitude goes to my co-advisor for his precious advice and guidance through the process of writing the thesis. My humble respect, goes to another co-advisor for his endless encouragement, intensive guidance who always provided constant support. Sincere thanks to all participants who spent their valuable time to join this study. Without their constant co-operation and help this research study could not have been completed. Finally, the authors would like to thank Christian Estacio for editing and revising the language of this manuscript.
References
- Ustün Y, Erdogan O, Esen E, Karsli ED. Comparison of the effects of 2 doses of methylprednisolone on pain, swelling, and trismus after third molar surgery. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2003;96:535–539. [CrossRef] [PubMed] [Google Scholar]
- McGrath C, Comfort MB, Lo EC, Luo Y. Changes in life quality following third molar surgery-the immediate postoperative period. Br dent J 2003;194:265–528. discussion 261. [CrossRef] [PubMed] [Google Scholar]
- Kim K, Brar P, Jakubowski J, Kaltman S, Lopez E. The use of corticosteroids and nonsteroidal antiinflammatory medication for the management of pain and inflammation after third molar surgery: a review of the literature. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2009;107:630–640. [CrossRef] [PubMed] [Google Scholar]
- Dhariwal DK, Goodey R, Shepherd JR. Trends in oral surgery in England and Wales 1991–2000. Br Dent J 2002;192:639–45. [CrossRef] [PubMed] [Google Scholar]
- Mercier P, Precious D. Risks and benefits of removal of impacted third molars. A critical review of the literature. Int J Oral Maxillofac Surg 1992;21:17–27. [CrossRef] [PubMed] [Google Scholar]
- Lim D, Ngeow WC. A Comparative study on the efficacy of submucosal injection of dexamethasone versus methylprednisolone in reducing postoperative sequelae after third molar surgery. J Oral Maxillofac Surg 2017;75:2278–2286. [CrossRef] [PubMed] [Google Scholar]
- Berge TI. Inability to work after surgical removal of mandibular third molars. Acta Odontol Scand 1997;55:64–69. [CrossRef] [PubMed] [Google Scholar]
- Göktay O, Satilmiş T, Garip H, Gönül O, Göker K. A comparison of the effects Of midazolam/fentanyl and midazolam/tramadol for conscious intravenous sedation during third molar extraction. J Oral Maxillofac Surg 2011;69:1594–1599. [CrossRef] [PubMed] [Google Scholar]
- Kaul RP, Godhi SS, Singh A. Autologous platelet rich plasma after third molar surgery: a comparative study. J Maxillofac Oral Surg 2012;11:200–205. [CrossRef] [PubMed] [Google Scholar]
- Koray M, Ofluoglu D, Onal EA, Ozgul M, Ersev H, Yaltirik M, et al. Efficacy of hyaluronic acid spray on swelling, pain, and trismus after surgical extraction of impacted mandibular third molars. Int J Oral Maxillofac Surg 2014;43:1399–1403. [CrossRef] [PubMed] [Google Scholar]
- Kumar N, Prasad K, Ramanujam L, Ranganath K, Dexith J, Chauhan A. Evaluation of treatment outcome after impacted mandibular third molar surgery with the use of autologous platelet-rich fibrin: a randomized controlled clinical study. J Oral Maxillofac Surg 2015;73:1042–1049. [CrossRef] [PubMed] [Google Scholar]
- Ozgul O, Senses F, Er N, Tekin U, Tuz HH, Alkan A, et al. Efficacy of platelet rich fibrin in the reduction of the pain and swelling after impacted third molar surgery: randomized multicenter split-mouth clinical trial. Head Face Med 2015;11:37. [CrossRef] [PubMed] [Google Scholar]
- Alissa R, Esposito M, Horner K, Oliver R. The influence of platelet-rich plasma on the healing of extraction sockets: an explorative randomised clinical trial. Eur J Oral Implantol 2010;3:121–134. [PubMed] [Google Scholar]
- Aloy-Prósper A, García-Mira B, Larrazabal-Morón C, Peñarrocha-Diago M. Distal probing depth and attachment level of lower second molars following surgical extraction of lower third molars: a literature review. Med Oral Patol Oral Cir Bucal 2010;15:e755–e759. [CrossRef] [PubMed] [Google Scholar]
- Gocmen G, Gonul O, Oktay NS, Yarat A, Goker K. The antioxidant and anti-inflammatory efficiency of hyaluronic acid after third molar extraction. J Craniomaxillofac Surg 2015;43:1033–1037. [CrossRef] [PubMed] [Google Scholar]
- Gönül O, Satılmış T, Bayram F, Göçmen G, Sipahi A, Göker K. Effect of submucosal application of tramadol on postoperative pain after third molar surgery. Head Face Med 2015;11:35. [CrossRef] [PubMed] [Google Scholar]
- Gönül O, Satilmiş T, Ciftci A, Sipahi A, Garip H, Göker K. Comparison of the effects of topical ketamine and tramadol on postoperative pain after mandibular molar extraction. J Oral Maxillofac Surg 2015;73:2103–2107. [CrossRef] [PubMed] [Google Scholar]
- van der Goes MC, Jacobs JW, Bijlsma JW. The value of glucocorticoid co-therapy in different rheumatic diseases–positive and adverse effects. Arthritis Res Ther 2014;16:S2. [CrossRef] [PubMed] [Google Scholar]
- Lutolf MP, Hubbell JA. Synthetic biomaterials as instructive extracellular microenvironments for morphogenesis in tissue engineering. Nat Biotechnol 2005;23:47–55. [CrossRef] [PubMed] [Google Scholar]
- Prestwich GD. Hyaluronic acid-based clinical biomaterials derived for cell and moleculedelivery in regenerative medicine. J Control Release 2011;155:193–199. [CrossRef] [PubMed] [Google Scholar]
- Rice KG. The chemistry, biology, and medical applications of hyaluronan and its derivatives. Ed Laurent TC J Med Chem 1998;41:5119–5336. [Google Scholar]
- Laurent TC, Fraser JR. Hyaluronan. FASEB J 1992;6:2397–404. [CrossRef] [PubMed] [Google Scholar]
- Pagnacco A, Vangelisti R, Erra C, Poma A. Double‑blind clinical trial versus placebo of a new sodium-hyaluronate-based gingival gel. Attual Ter in 1997;15:1–7. [Google Scholar]
- Dahiya P, Kamal R. Hyaluronic acid: a boon in periodontal therapy. N Am J Med Sci 2013;5:309–315. [CrossRef] [PubMed] [Google Scholar]
- Møystad A, Mork-Knutsen BB, Bjørnland T. Injection of sodium hyaluronate compared to a corticosteroid in the treatment of patients with temporo mandibular joint osteoarthritis: a CT evaluation. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2008;105:e53–60. [CrossRef] [Google Scholar]
- Shuborna NS, Chaiyasamut T, Sakdajeyont W, Vorakulpipat C, Rojvanakarn M, Wongsirichat N. Generation of novel hyaluronic acid biomaterials for study of pain in third molar intervention: a review. J Dent Anesth Pain Med 2019;19:11–19. [CrossRef] [PubMed] [Google Scholar]
- Sato R, Yamamoto H, Kasai K, Yamauchi M. Distribution pattern of versican, link protein and hyaluronic acid in the rat jreiodontal ligament during exjreimental tooth movement. J Periodont Res 2002;37:15–22. [CrossRef] [Google Scholar]
- Gontiya G, Galgali SR. Effect of hyaluronan on periodontitis: a clinical and histological study. J Indian Soc Periodontol 2012;16:184–192. [CrossRef] [PubMed] [Google Scholar]
- Migliore A, Granata M. Intra-articular use of hyaluronic acid in the treatment of osteoarthritis. Clin Interv Aging 2008;3:365–369. [CrossRef] [Google Scholar]
- Allegra L, Della Patrona S, Petrigni G. Hyaluronic acid: perspectives in lung diseases. Handb Exp Pharmacol 2012;385–401. [CrossRef] [Google Scholar]
- Yilmaz N, Demirtas N, Kazancioglu HO, Bayer S, Acar AH, Mihmanli A. The efficacy of hyaluronic acid in postextraction sockets of impacted third molars: a pilot study. Niger J Clin Pract 2017;20:1626–1631. [PubMed] [Google Scholar]
- Bayoum A, Nadershah M, Albandar A, Alsulaimani B, Sankour I, Gadi L, et al. The effect of cross-linked hyaluronic acid in surgical extraction of impacted mandibular third molars. Int J Dent Oral Health 2018;4:dx. [Google Scholar]
- Goiato MC, da Silva EVF, de Medeiros RA, Túrcio KH, Dos Santos DM. Are intra-articular injections of hyaluronic acid effective for the treatment of temporomandibular disorders? Int J Oral Maxillofac Surg 2016;45:1531–1537. [CrossRef] [PubMed] [Google Scholar]
- Guarda-Nardini L, Stifano M, Brombin C, Salmaso L, Manfredini D. A one-year case series of arthrocentesis with hyaluronic acid injections for temporomandibular joint osteoarthritis. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2007;103:e14–22. [CrossRef] [PubMed] [Google Scholar]
- Fonseca RDMEFB, Januzzi E, Ferreira LA, Grossmann E, Carvalho ACP, de Oliveira PG, et al. Effectiveness of sequential viscosupplementation in temporomandibular joint internal derangements and symptomatology: a case series. Pain Res Manag 2018;2018:5392538. [PubMed] [Google Scholar]
- Gurung T, Singh RK, Mohammad S, Pal US, Mahdi AA, Kumar M. Efficacy of arthrocentesis versus arthrocentesis with sodium hyaluronic acid in temporomandibular joint osteoarthritis: a comparison. Natl J Maxillofac Surg 2017 Jan-Jun;8:41–49. [CrossRef] [PubMed] [Google Scholar]
- Ferreira N, Masterson D, Lopes de Lima R, de Souza Moura B, Oliveira AT, da Silva Fidalgo TK, et al. Efficacy of viscosupplementation with hyaluronic acid in temporomandibular disorders: a systematic review. J Craniomaxillofacial Surg 2018;46:1943–1952. [CrossRef] [Google Scholar]
- Bjørnland T, Gjaerum AA, Møystad A. Osteoarthritis of the temporomandibular joint: an evaluation of the effects and complications of corticosteroid injection compared with injection with sodium hyaluronate. J Oral Rehabil 2007;34:583–589. [CrossRef] [PubMed] [Google Scholar]
- Bali A, Bali D, Sharma A, Verma G. Is Pederson index a true predictive difficulty index for impacted mandibular third molar surgery? A meta-analysis. J Maxillofac Oral Surg. 10.1007/s12663-012-0435-x [PubMed] [Google Scholar]
- Latt MM, Kiattavorncharoen S, Boonsiriseth K, Pairuchvej V, Wongsirichat N. The efficacy of dexamethasone injection on postoperative pain in lower third molar surgery. J Dent Anesth Pain Med 2016;16:95–102. [CrossRef] [PubMed] [Google Scholar]
- Schultze-Mosgau S, Schmelzeisen R, Frölich JC, Schmele H. Use of ibuprofen and methylprednisolone for the prevention of pain and swelling after removal of impacted third molars. J Oral Maxillofac Surg 1995;53:2–7; discussion 7–8. [CrossRef] [PubMed] [Google Scholar]
- Kopp S, Wenneberg B, Haraldson T, Carlsson GE. The short-term effect of intra-articular injections of sodium hyaluronate and corticosteroid on temporomandibular joint pain and dysfunction. J Oral Maxillofac Surg 1985;43:429–435. [CrossRef] [PubMed] [Google Scholar]
- Merchant R, Khalid I, Natrajan S, Galinde J. Comparative evaluation of clinical efficacy of hyaluronic acid spray versus normal saline spray on swelling, pain, and trimus after surgical extraction of impacted mandibular third molar − a randomized controlled split mouth study. IJSR 2018;7:152–159. [Google Scholar]
- Yura S, Totsuka Y, Yoshikawa T, Inoue N. Can arthrocentesis release intracapsular adhesions? Arthroscopic findings before and after irrigation under sufficient hydraulic pressure. J Oral Maxillofac Surg 2003;61:1253–1256. [CrossRef] [PubMed] [Google Scholar]
- Afat I.M, Akdoğan ET, Gönül O. Effects of Leukocyte- and platelet- rich fibrin alone and combined with hyaluronic acid on pain, edema, and trismus after surgical extraction of impacted mandibular third molars. J Oral Maxillofac Surg 2018;76:926–932. [CrossRef] [PubMed] [Google Scholar]
- Lee JH, Jung JY, Bang D. The efficacy of topical 0.2% hyaluronic acid gel on recurrent oral ulcers: comparison between recurrent aphthous ulcers and the oral ulcers of Behçet's disease. J Eur Acad Dermatol Venereol 2008;22:590–595. [CrossRef] [PubMed] [Google Scholar]
- Romeo U, Libotte F, Palaia G, Galanakis A, Gaimari G, Tenore G, et al. Oral soft tissue wound healing after laser surgery with or without a pool of amino acids and sodium hyaluronate: a randomized clinical study. Photomed Laser Surg 2014;32:10–16. [CrossRef] [PubMed] [Google Scholar]
- Chang YL, Lo YJ, Feng SW, Huang YC, Tsai HY, Lin CT, et al. Bone healing improvements using hyaluronic acid and hydroxyapatite/beta-tricalcium phosphate in combination: an animal study. Biomed Res Int 2016;2016:8301624. [PubMed] [Google Scholar]
- Grond S, Sablotzki A. Clinical pharmacology of tramadol. Clin Pharmacokinet 2004;43:879–923. [CrossRef] [PubMed] [Google Scholar]
All Tables
All Figures
 |
Fig. 1 Male-Female ratio in this study who assessed pre and post-operatively all 3 clinical parameters. |
| In the text | |
 |
Fig. 2 (A) Hyalgun (Hyaluronic acid solution 20 mg/2 ml), (B) Gelfoam, (C) Hyaluronic acid solution applied with gelfoam. |
| In the text | |
 |
Fig. 3 Facial swelling and trismus measurements. A: Gonion to lateral canthus of eye; B: Tragus to angle of mouth; C: Tragus to pogonion; D: Maximum inter-incisal. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


