| Issue |
J Oral Med Oral Surg
Volume 26, Number 2, 2020
|
|
|---|---|---|
| Article Number | 21 | |
| Number of page(s) | 3 | |
| Section | Cas clinique et revue de la littérature / Up-to date review and case report | |
| DOI | https://doi.org/10.1051/mbcb/2020017 | |
| Published online | 05 June 2020 | |
Up-to Date Review And Case Report
Saprochaete capitata oropharyngeal infection in a neutropenic patient: unusual presentation
1
Parasitology-Mycology Department of the Moulay Ismail Military Hospital, Meknes, Morocco
2
Microbiology Department of the Moulay Ismail Military Hospital, Meknes, Morocco
3
Hematology Department of the Moulay Ismail Military Hospital, Meknes, Morocco
* Correspondence: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
4
March
2020
Accepted:
21
April
2020
Abstract
Introduction: Saprochete capitata is unusual etiologic agent in immunocompromised patients, particularly in those with hematologic malignancy and severe neutropenia. Most often, infections of the oral cavity are manifested clinically as oral candidiasis. Invasive forms are rarely described. Observation: a 63-year-old man consulted for pseudomembranous lesions associated with ulcero perforating lesion of the tongue and palatal region ulcerations. All evolving in a context of profound physical deterioration and severe neutropenia. Mycological examination showed Saprochaete capitata. The evolution was favorable with oral voriconazole. Comment: Saprochaete capitata invasive fungal infections have become an important cause of morbidity and mortality, particularly in hematology-oncology patients. Invasive or non-invasive, oropharyngeal involvement with this pathogen should not be underestimated in the neutropenic patient. They are the main starting point for fongemia of this pathogen, which is often fatal. Conclusion: Saprochete capitata is now recognized emerging etiologic agent in patients with hematological malignancy and severe neutropenia. Early detection and diagnosis of these fungal infections could lead to reduced morbidity and mortality, particularly in locally invasive infection.
Key words: Saprochaete capitata / oral mycosis / Voriconazole
© The authors, 2020
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
Saprochaete capitata (S. capitata), formerly known as Geotrichum capitatum, is an ascomycete fungus which has subject of several taxonomic revisions in recent decades [1]. It is a cosmopolitan yeast and a usual contaminant found in the environment and dairy products. As it is part of the normal flora of the skin, bronchi, mouth and the gastrointestinal tract in about 30% of the general population [2]. S. capitata Infections are very common and occur almost exclusively in immunocompromised patients, particularly those with hematologic malignancy and severe neutropenia [3,4].
We report a rare case of S. capitata invasive oropharyngeal infection in a neutropenic patient, through which will describe the clinical, biological and therapeutic aspect of this infection and comparison with published data.
A 63 year old man, non-alcohol-smoking, was admitted for fever, chills, headache and dysphagia. Oral cavity examination showed pseudomembranous lesions associated with ulcero perforating lesion of the tongue and ulcerations of the palatal region. All evolving in a context of profound physical deterioration and weight loss estimated at 7 kg. Empirical therapy combining fluconazole, ceftazidime and vancomycin has been initiated. The patient's history dates back to June 2018 when he was treated for chronic lymphocytic leukemia whose response was complete after a treatment protocol combining rituximab, fludarabine and cyclophosphamide.
Clinical examination showed no lymphadenopathy or organomegaly. The thoracic scanner was without particularity. Blood test showed CRP at 49 mg/L, normochromic normocytic non-regenerative anemia (hemoglobin at 7.5 g/dL), thrombocytopenia at 48 × 109/L, neutropenia at 0.64 × 109/L, and lymphopenia at 0.14 × 109/L. The myelogram, which found a bone-marrow erythroblastosis estimated at 60%, multiline dysplasia and some images of hemophagocytosis, evoked a myelodysplastic syndrome probably secondary to therapeutic toxicity. All blood cultures returned negative.
Direct examination realized on lesions swab objectified the presence of numerous septate mycelial filaments with dichotomous ramifications and secondary filaments disposed at acute angles. Rare arthrospores have been observed (Fig. 1). Culture on Sabouraud-Chloramphenicol medium agar at 37 °C allowed whitish, dry, wrinkled, finely and fluffy colonies after 48 hours (Fig. 2). Microscopic examination revealed arthroconidia and annelloconidia (Fig. 3). On the Api 20C Aux system (Biomerieux, France), the isolate assimilated only D-glucose and D-galactose. The urease test was negative. These morphological and biochemical characteristics were compatible with that of S. capitata. The sequencing of the ITS1 and ITS2 regions for a more formal identification of the species was not realized due to lack of means.
At day 7 of treatment, the patient showed no signs of clinical improvement. Antifungal therapy was changed for oral voriconazole 200 mg twice daily and continued for 3 weeks. The evolution was favorable with disappearance of fever and complete regression of oral cavity lesions (Fig. 4). The patient was followed on an outpatient basis to explore his pancytopenia.
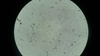 |
Fig. 1 Direct examination on lesions swab (×400). Numerous septate mycelial filaments with dichotomous ramifications and secondary filaments disposed at acute angles. Rare arthrospores. |
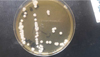 |
Fig. 2 Macroscopic image of Saprochaete capitata. 48-hour colonies grown on Sabouraud-Chloramphenicol agar medium. |
 |
Fig. 3 Microscopic image of Saprochaete capitata colony (×400). Wet-mount preparations showing arthroconidia and annelloconidia. |
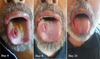 |
Fig. 4 Clinical course of tongue lesions after initiation of oral voriconazole. |
Comment
S. capitata (teleomorph Magnusiomyces capitatus) is an ascomycota fungus belonging to the Saccharomycetes class, Dipodascaceae family. Given a new in-depth taxonomic assessment based on the study of ribosomal structure, it has been reclassified in the Saprochaete genus (instead of Geotrichum) [1]. It is a negative urease yeast, non-fermenting and assimilates glucose and galactose. On blood agar medium and Sabouraud Dextrose Agar, it gives white to cream colonies, and can grow at 45 °C. and in presence of cycloheximide [5].
Over the past two decades, the incidence of invasive fungal infections (IFI) has increased considerably. They have become an important cause of morbidity and mortality, particularly in patients in hematology-oncology [6]. Infections with S. capitata are among the rarest. According to an international antifungal surveillance program led by ARTEMIS DISK, the prevalence of S.capitata was 1% among 11,240 non-Candida yeast isolates examined between 1997 and 2007 [7]. A multicenter study involving 589 adult and pediatric patients with malignant hemopathies, the incidence of IFI was 4.6%, and the prevalence of S. capitata among all isolates in adult patients was 6.2% [3]. Similarly, this is the second case of IFI reported to S. capitata [8] and the first case occurring in a patient with a malignant hemopathy in our institution.
According to previous S. capitata IFI reports, fungal infections and pulmonary infections were among the most frequent [3,4]. Indeed, fungal infections of the oral cavity S. capitata occurred most often in an array of superficial mucosal involvement similar to that of oral candidiasis [9–11]. The most aggressive damage that resembled of mucormycosis and aspergillosis ulceration has been rarely reported [12,9]. Invasive or non-invasive, oropharyngeal involvement with this pathogen should not be underestimated in the neutropenic patient, Özdemir ZC et al. reported three cases of fatal fungemia at this yeast in patients of acute lymphoblastic leukemia whose starting point was an oral fungal infection [13]. In addition to clinical context of this patient, the invasive character was established in front of the soft tissue involvement illustrated by the tongue perforating lesion and the results of the mycological examination. In addition, fungemia was not excluded due to the very moderate sensitivity of the blood culture.
At present, the optimal antifungal treatments proposed are still unclear due to the rarity of this etiological agent and few published studies. A review of the literature suggests intrinsic resistance to echinocandins and moderate sensitivity to fluconazole and itraconazole [14,15]. However, clinical success has been reported with amphotericin B with or without 5-fluorocytosine [9,16,17]. Similarly, voriconazole can also be used alone or in combination with amphotericin B [18]. In this patient, treatment with oral voriconazole 200 mg twice daily for 3 weeks was successful, shown by the disappearance of the fever and complete regression of oropharyngeal lesions.
Conclusion
S. capitata is now recognized as an emerging etiological agent in patients with malignant hemopathy and severe neutropenia. Early detection and diagnosis of these fungal infections could lead to reduced morbidity and mortality, particularly in the case of locally invasive infection [11]. The morphological characteristics observed on direct examination and on stained smears can allow an early preliminary diagnosis, thus significantly reducing the time required for the establishment of an appropriate antifungal treatment [19].
Conflicts of interests
The authors declare that they have no conflicts of interest in relation to this article.
References
- De Hoog GS, Smith MT. Ribosomal gene phylogeny and species delimitation in Geotrichum and its telemorphs. Stud Mycol 2004;50:489–515. [Google Scholar]
- Pottier I, Gente S, Vernoux JP, Guéguen M. Safety assessment of dairy microorganisms: Geotrichum candidum . Int J Food Microbiol 2008;126:327–332. [CrossRef] [PubMed] [Google Scholar]
- Montagna MT, Giglio OD, Napoli C, Lovero G, Caggiano G, Delia M, et al. Invasive fungal infections in patients with hematologic malignancies (Aurora Project): lights and shadows during 18-months surveillance. Int J Mol Sci 2012;13:774–787. [Google Scholar]
- Durán Graeff L, Seidel D, Vehreschild MJGT, Hamprecht A, Kindo A, Racil Z, et al. Invasive infections due to Saprochaete and Geotrichum species: report of 23 cases from the FungiScope Registry. Mycoses 2017;60:273–279. [CrossRef] [PubMed] [Google Scholar]
- Freydiere A-M, Guinet R, Boiron P. Yeast identification in the clinical microbiology laboratory: phenotypical methods. Med Mycol 2001;39:9–33. [Google Scholar]
- Richardson M, Lass-Flörl C. Changing epidemiology of systemic fungal infections. Clin Microbiol Infect 2008;14:5–24. [Google Scholar]
- Pfaller MA, Diekema DJ, Gibbs DL, Newell VA, Ellis D, Tullio V, et al. Results from the ARTEMIS DISK global antifungal surveillance study, 1997 to 2007: a 10.5-year analysis of susceptibilities of Candida species to fluconazole and voriconazole as determined by CLSI standardized disk diffusion. J Clin Microbiol 2010;48:1366–1377. [PubMed] [Google Scholar]
- El-Hassani I, Deham H, Touaoussa A, Er-rami M. Infection pulmonaire à Geotrichum capitatum à propos d'un cas et revue de la littérature. J Mycol Med 2010;24:341–344. [Google Scholar]
- Bonifaz A, Vázquez-González D, Macías B, Paredes-Farrera F, Hernández MA, Araiza J, et al. Oral geotrichosis: report of 12 cases. J Oral Sci 2010;52:477–483. [CrossRef] [PubMed] [Google Scholar]
- Bonifaz A, Armas-Vázquez A, Ponce-Olivera RM. Infección por Saprochaete capitata en paciente inmunosuprimido. Dermatol Rev Mex 2014;58:380–384. [Google Scholar]
- Telles DR, Karki N, Marshall MW. Oral fungal infections: diagnosis and management. Dent Clin North Am. 2017;61:319–349. [CrossRef] [PubMed] [Google Scholar]
- Listemann H, Schönrock-Nabulsi P, Kuse R, Meigel W. Geotrichosis of oral mucosa. Mycoses 1996;39:289–291. [CrossRef] [PubMed] [Google Scholar]
- Özdemir ZC, Bozkurt Turhan A, Düzenli Kar Y, Dinleyici ÇE, Bör Ö. Fatal course of Saprochaete capitata fungemia in children with acute lymphoblastic leukemia. Oncol 2017;34:66–72. [Google Scholar]
- Arendrup MC, Boekhout T, Akova M, et al. ESCMID and ECMM joint clinical guidelines for the diagnosis and management of rare invasive yeast infections. Clin Microbiol Infect 2014;20:76–98. [Google Scholar]
- Girmenia C, Pizzarelli G, D'Antonio D, Cristini F, Martino P. In vitro susceptibility testing of Geotrichum capitatum: comparison of the E-Test, disk diffusion, and sensititre colorimetric methods with the NCCLS M27-A2 broth microdilution reference method. Antimicrob Agents Chemother 2003;47:3985–3988. [CrossRef] [PubMed] [Google Scholar]
- Gadea I, Cuenca-Estrella M, Prieto E, Diaz-Guerra TM, Garcia-Cia JI, Mellado E, et al. Genotyping and antifungal susceptibility profile of Dipodascus capitatus isolates causing disseminated infection in seven hematological patients of a tertiary hospital. J Clin Microbiol 2004;42:1832–1836. [PubMed] [Google Scholar]
- Hajar Z, Medawar W, Rizk N. Saprochaete capitata (Geotrichum capitatum), an emerging fungal infection in kidney transplant recipients. J Mycol Med 2018;28:387–389. [CrossRef] [PubMed] [Google Scholar]
- Birrenbach T, Bertschy S, Aebersold F, Mueller NJ, Achermann Y, Muehlethaler K, et al. Emergence of Blastoschizomyces capitatus yeast infections, Central Europe. Emerg Infect Dis 2012;18:98–101. [CrossRef] [PubMed] [Google Scholar]
- Alobaid K, Abdullah AA, Ahmad S, Joseph L, Khan Z. Magnusiomyces capitatus fungemia: the value of direct microscopy in early diagnosis. Med Mycol Case Rep 2019;25:32–34. [PubMed] [Google Scholar]
All Figures
 |
Fig. 1 Direct examination on lesions swab (×400). Numerous septate mycelial filaments with dichotomous ramifications and secondary filaments disposed at acute angles. Rare arthrospores. |
| In the text | |
 |
Fig. 2 Macroscopic image of Saprochaete capitata. 48-hour colonies grown on Sabouraud-Chloramphenicol agar medium. |
| In the text | |
 |
Fig. 3 Microscopic image of Saprochaete capitata colony (×400). Wet-mount preparations showing arthroconidia and annelloconidia. |
| In the text | |
 |
Fig. 4 Clinical course of tongue lesions after initiation of oral voriconazole. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


