| Issue |
Med Buccale Chir Buccale
Volume 23, Number 3, October 2017
|
|
|---|---|---|
| Page(s) | 156 - 159 | |
| Section | Cas clinique et revue de la littérature / Up-to date review and case report | |
| DOI | https://doi.org/10.1051/mbcb/2017003 | |
| Published online | 27 October 2017 | |
Up-to Date Review And Case Report
Differential diagnosis of an oral mucosal pigmented lesion: a case of essential melanosis
1
CHU Clermont-Ferrand, Service d'Odontologie,
F-63003
Clermont-Ferrand, France
2
CHU Clermont-Ferrand, Service de Stomatologie, Chirurgie maxillo-faciale et chirurgie plastique de la face,
F-63003
Clermont-Ferrand, France
3
Université Clermont Auvergne, BP 10448, F-63000 Clermont-Ferrand & Inserm, Neuro-Dol,
63001
Clermont-Ferrand, France
* Correspondence: This email address is being protected from spambots. You need JavaScript enabled to view it.
Received:
19
October
2015
Accepted:
8
February
2017
Abstract
Introduction: The presence of a pigmented lesion of the oral mucosa raises numerous diagnostic hypotheses. The INCa (National Cancer Institute) considers “any pigmented lesion of the oral mucosa as a melanoma until proven otherwise.” A histological confirmation is thus necessary. Clinical observation: A 64-year-old female patient consulted for a pigmented lesion of the marginal gingiva of 11 and 21, corresponding to a single round macule of approximately 0.5 cm diameter, brown, homogeneous, covered with a slight keratinization, with regular edges. The rest of the clinical examination was unremarkable. An excisional biopsy confirmed the diagnosis of essential melanosis or Laugier–Hunziker syndrome. Discussion: Pigmented lesions may occur in an isolated form or may be a component of major syndromes. Unique pigmentation of the oral mucosa can be of endogenous origin (e.g., malignant melanoma, essential melanosis, lentigo, different nevi) or of exogenous origin (e.g., ethnic pigmentations, metallic or medicinal origin, vascular lesions, and neuroectodermal tumor in children). Conclusion: Although rare at the gingival level, essential melanosis must be considered in the differential diagnosis of a single flat lesion. Nevertheless, melanoma should always be ruled out, and its early diagnosis is the only favorable prognostic factor in its evolution.
Key words: pigmented lesion / Laugier–Hunziker syndrome / melanoma
© The authors, 2017
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Introduction
The National Cancer Institute considers “any pigmented lesion on the oral mucosa as a melanoma, until evidence to the contrary.” In the face of the number of possible differential diagnoses and their similarities, it is necessary to regularly perform histological examinations of any pigmented lesion of the oral mucosa to confirm or disprove the clinically suspected diagnostic hypothesis and to formally exclude melanoma.
Clinical observation
A 64-year-old patient, a nurse of Caucasian origin, arrived for consultation for an oral mucosal pathology for a brownish lesion located on the marginal gingiva next to the crowns of the upper central incisors. This asymptomatic lesion appeared 2 years before the consultation, and had not increased in size over time. Its unsightly appearance prompted her consultation.
The patient did not have any relevant personal or family history and did not have allergies. She was a nonsmoker and did not consume alcohol regularly. She was operated for a rotator cuff rupture of the right shoulder and a bunion on the left foot.
At clinical examination, her oral condition was satisfactory, and there was no clinical evidence of oral or systemic pathology. General extraoral examination (particularly of the skin, appendages, and other mucous membranes) was normal, similar to local and regional examinations. Palpation revealed no enlarged lymph nodes, and the rest of the oral cavity was unremarkable.
The pigmented lesion corresponded to a single round macule of approximately 0.5 cm in diameter, brown, uniform, covered with a slight keratosis, with regular edges (Fig. 1). At palpation, the lesion was soft and nontender.
In the face of this single, brownish gingival lesion, a nevocellular nevus of keratotic appearance was initially suspected. Melanoma, although unlikely, was nevertheless considered as a differential diagnosis.
An excisional biopsy was immediately performed because of the small size of the mucosal lesion for get a histopathological examination to confirm or deny the clinical diagnosis.
The results of the histopathological examination were as follows: squamous mucosa without architectural anomalies, a hypermelanosis at the level of the basal layers without significant lentiginous hyperplasia associated with pigmentary incontinence. The appearance favored a simple melanic macule (Fig. 2).
The final diagnosis confirmed an essential melanosis or Laugier–Hunziker syndrome of a gingival location, with a complete excision (Fig. 3). The patient was informed of the result.
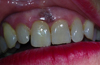 |
Fig. 1 Intraoral photography of an essential melanosis or Laugier–Hunziker syndrome of the attached gingiva at the neck of the teeth 11 and 21. |
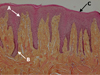 |
Fig. 2 Histological section of the essential melanosis or Laugier–Hunziker syndrome (HES stain, ×10). Papillomatosis (A), melanin hyperpigmentation of the basal layer (B), and a surface keratosis (C) may be noted. There is no melanocyte proliferation. |
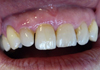 |
Fig. 3 Intraoral photography after 3 months of healing indicating the restoration of normalcy. |
Discussion
The main difficulty while treating a single pigmented lesion of the oral mucosa is to eliminate other differential diagnoses, especially malignant melanoma.
A single pigmented macule of the oral mucosa can be of endogenous or exogenous origin.
Endogenous causes are melanoma, lentigo, essential melanosis or Laugier–Hunziker syndrome, nevocellular nevus, blue nevus, and melanoacanthoma. Exogenous causes are ethnic, medical, or metallic tattoos; hemangiomas; Kaposi's disease; and, in children, neuroectodermal tumor [1–3].
Clinical features of melanoma should be systematically assessed by a thorough clinical and histological examination.
At medical history taking, we inquire the age of the lesion: recent or old, personal and family history of melanoma or other skin diseases in particular, the existence of associated pathologies (skin diseases, syndromes, endocrine, metabolic, vascular diseases), medication (synthetic antimalarials, quinidine, minocycline, busulfan, etc.), patient profession and ethnicity, the existence of exogenous causes.
At intraoral clinical examination, the lesion is described precisely: its location; number (single, multiple, or diffuse); type (macule, papule, nodule); infiltration; color; homogeneity; and extension to the rest of the oral cavity, other mucous membranes, skin, and appendages. Cervical lymph node areas should be systematically palpated to rule out lymphadenopathy.
To ensure that it is not a melanoma, a biopsy, specifically an excisional biopsy (depending on the size of the lesion), is performed to have histological confirmation of the nature of the lesion. Fontana–Masson staining during the histological examination allows the differentiation of pigmentation of exogenous origin (negative staining) from pigmentation of endogenous origin of melanin nature (positive staining). The histological examination also confirms the nature of the lesion. For example, hypermelanocytosis (increase in the number of melanocytes) is found in lentigo, hypermelaninosis (increase in the number of melanin pigments) in essential melanosis, and nevocyte clusters (cluster of 5–20 cells) in nevi.
Essential melanosis or Laugier–Hunziker syndrome essentially affects middle-aged, Caucasian women. This condition is rare, has a benign evolution, and its etiology remains unknown. It clinically manifests as brown or black, homogeneous macules, well defined and small in size. These lesions may be isolated at the level of the anterior part of the oral cavity or associated with cutaneous, genital, or anal macules. Longitudinal melanonychia can be similarly found, and in exceptional cases, conjunctival or esophageal macules [4,5].
Histology, although nonspecific, found hypermelaninosis. The basal layer is hyperpigmented and sometimes acanthotic. There is a migration of the melanin pigment in the superficial layer which is sometimes picked up by the melanophages. The number, morphology, and distribution of melanocytes are not affected and as such there is no melanocyte proliferation.
Melanoma is rare in the oral mucosa (< 1% melanomas). It represents 0.5% of all cancers of the oral cavity and most often affects black and Asian subjects and men aged 40–70 years [6]. It is characterized by macules of varying color − brownish, black, or bluish − with irregular edges or may still be manifested in nodular form. Melanoma appears on the palate in 50% cases and on the gums (essentially superior) in 20% cases. It is asymptomatic at an early stage and its diagnosis is most often made incidentally − e.g., during oral cavity examination for dental care. At an advanced stage (because of a delayed diagnosis), it appears as a symptomatic ulcerated, bleeding tumor nodule with the presence of lymphadenopathy, and is sometimes associated with loss of teeth because of the destruction of the underlying bone. The criteria used to describe a pigmented lesion are the following: A (asymmetry), B (borders or edges), C (color), D (diameter), E (evolution). An asymmetrical lesion, of irregular edges, blackish dark color, diameter > 6 mm, which rapidly increases in size points toward a melanoma diagnosis.
Its excision at an early stage is recommended to avoid its development into a more evolved form and its metastatic extension, as melanoma has a high metastatic power into the central nervous system and other viscera, especially in an environment like the oral mucosa, which is highly vascular.
Histological analysis reveals a proliferation of tumor cells containing melanin pigments grouped in clusters or in thecas. In contrast to cutaneous melanomas, the Breslow index and the Clark classification are not evaluated for mucosal melanomas.
Fontana coloring allows the visualization of melanin pigments. Similarly, the S100 protein and the antigen HMB45 can be evaluated in nonpigmented forms or in doubtful cases.
An assessment of the extension according to findings on brain, cervical, and thoracic-abdominal computed tomography scan is proposed to evaluate the depth and surface extension and the search of metastases. PET is another alternative. Treatment is based on the widest possible excision, possibly with associated nodal dissection depending on the tumor extent. Radiotherapy, chemotherapy, and immunotherapy are used depending on the physician's discretion as complementary treatment or in cases where surgery is contraindicated (bad general condition of the patient, extensive tumor spread).
Its prognosis is dark and more damaging than on the skin because of late diagnosis, the vascularity of the oral mucosa (which promotes metastatic extension), and the anatomical difficulty of wide excision. The 5-year survival rate is estimated between 10% and 20% [7,8] with an average survival of 25 months [9].
Malignant lentigo is a melanocytic tumor often considered to be a melanoma in situ (Dubreuilh's melanoma). Its extension remains superficial and is epidermal; however, malignant lentigo can progress into a melanoma in 30–40% cases (invasion of the dermis by cancerous melanocytes).
Clinically, it is a pigmented macule of brown or black color, asymmetrical, most often found on the parts of the body exposed to the sun (back of the hands), the face and the neck. Biopsy or excisional biopsy will be necessary of at the outset (depending on its size and location) to obtain histological diagnostic confirmation. Intense or prolonged sun exposure is a risk factor.
From a histological point of view, malignant lentigo is characterized by a proliferation of atypical melanocytes to the dermoepidermal junction. The differential diagnoses to consider with malignant lentigo are the following: solar lentigo, seborrheic keratosis, keratotic lichen planus, actinic keratosis, and melanocytic nevus.
Its treatment is based on surgical excision with histological examination. As a second line of treatment, topical imiquimod chemotherapy and radiotherapy can be used [10].
Lentigo, still called unique melanotic macule, is common in adults and is of a benign nature. It manifests in the form of a brown or blackish macule that can measure 0.5–1 cm in diameter. It can affect the skin and mucous membranes. The usual location in the oral cavity is the lower lip. In addition, in patients with dark skin, this lesion can be found on the tongue or the palate [11–13]. When these macules are multiple, we talk about lentiginosis. The latter can be part of more complex syndromes such as LEOPARD's syndrome, Touraine's neurodystrophic lentiginosis, Peutz–Jeghers' syndrome [14] or even Carney's syndrome. The histological examination finds epithelial hyperplasia, acanthosis, melanic pigmentation of the basal layers of the epithelium (hypermelanocytosis), and many melanophages in the underlying layers. There is no regrouping in thecas.
Nevocellular nevus is infrequent in the oral mucosa and more often affect subjects aged 30–40 years. It is frequently located on the palate, to the inside of the cheeks and sometimes gum and the labial mucosa in the form of a macule with a size 0.3–1 cm, a brown to dark blue hue, and a homogeneous experience. It can also manifest in the form of a nodule.
Clinical distinction is difficult between a nevocellular nevus and melanoma, thus the need for histological analysis [15].
Histological analysis found a proliferation of nevus cells, regular, rounded, grouped in thecas or layers within the basal membrane and the lamina propria.
Surgical excision is the rule to rule out early melanomas or to prevent their evolution.
Blue nevus is rare in the oral mucosa, and is most often located on the palate. It is most often found in children and young adults, and in women. It has bluish gray color, and can be macular or nodular. Fusiform cells located exclusively in the lamina propria can be found on the histological sections. The deep location of these cells explains the bluish coloration of the nevus. Blue nevus generally does not degenerate, but its resemblance to melanoma is an indication for surgical excision.
Melanoacanthoma is exceptionally rare in the oral mucosa and is more often found on the palate and inner cheeks. It affects young adults and most frequently women with black skin. Local trauma or a history of chronic irritation are triggering factors. It can measure up to several centimeters in diameter.
The pigmentation of exogenous origin can be either accidental or voluntary. Pigmentation of accidental causes may be due to tar, pigmentation due to the metals present in the fillings or crowns. Sometimes a mucosal break with intramucosal deposit of dental material may be observed.
Pigmentation of voluntary origin may be because of ethnic pigmentations in African tribes or because of tattoos with the presence of pigmented scar on cheeks or the buccal gingiva [16].
Pigmentation of vascular origin is dark red or purplish, close to black. These lesions are represented by bruises, angiomas plans, and Kaposi's disease. The vascular nature of the lesion manifests as diurnal changes in angioma volumes, the position of the head, the heat and by their blanching to the pressure.
In Kaposi's disease, the lesion is macular or nodular, with dark purplish coloring, and diffuse location, with a possible extension to the whole body.
Conclusion
Essential melanosis or Laugier–Hunziker syndrome belongs to the group of single pigmented macules occurring in adults. Although rare, it is necessary to know how to clinically diagnose when encountering a single, flat pigmented lesion of the oral cavity, particularly in women of Caucasian origin.
Even if clinical suspicion is strong, the only way for a final diagnosis of a pigmented oral lesion remains the histopathological examination.
Therefore, it is necessary to reinforce the importance of a comprehensive intraoral examination during any consultation with the dental surgeon, complemented by full dermatological examination if there is a mucosal lesion as required. Early detection of melanoma and prompt treatment allows for a decrease the morbidity and mortality of this pathology.
Conflicts of interests
The authors declare that they have no conflicts of interest in relation to this article.
References
- Brocheriou C, Kuffer R, Verola O. Lésions pigmentées de la cavité buccale. Ann Pathol 1995;5:221–229. [EDP Sciences] [Google Scholar]
- Ciçek Y, Ertas U. The normal and pathological pigmentation of oral mucous membrane: a review. J Contemp Dent Pract 2003;4:76–86. [Google Scholar]
- Popa C, Stelea C, Popa R, Popescu E. Oral and perioral endogenous pigmented lesions. Rev Med Chir Soc Med Nat Lasi 2008;112:1054–1060. [Google Scholar]
- Rangwala S, Doherty CB, Katta R. Laugier-Hunziker syndrome: a case report and review of the literature. Dermatol Online J 2010;16:9. [Google Scholar]
- Lombardi T, Combremont F, Samson J. Mélanose essentielle ou syndrome de Laugier-Hunziker. Med Buccale Chir Buccale 2012;18:387–388. [CrossRef] [EDP Sciences] [Google Scholar]
- Patel SG, Prasad ML, Escrig M, Singh B, Shaha AR, Kraus DH, Boyle JO, Huvos AG, Busam K, Shah JP. Primary mucosal malignant melanoma of the head and neck. Head Neck 2002;24:247–257. [CrossRef] [PubMed] [Google Scholar]
- Eisen D, Voorhees JJ. Oral melanoma and other pigmented lesions of the oral cavity. J Am Acad Dermatol 1991;24:527–537. [CrossRef] [PubMed] [Google Scholar]
- Manolidis S, Donald PJ. Malignant mucosal melanoma of the head and neck. Review of the literature and report of 14 patients. Cancer 1997;80:1373–1386. [Google Scholar]
- Fricain JC, Meyer M, Catros S, Jouary S. Black is black! Med Buccale Chir Buccale 2010;16:191–192. [CrossRef] [EDP Sciences] [Google Scholar]
- Kallini JR, Jain SK, Khachemoune A. Lentigo maligna: review of salient characteristics and management. Am J Clin Dermatol 2013;14:473–480. [CrossRef] [PubMed] [Google Scholar]
- Cribier B et al. Did you say lentigo? Ann Dermatol Venereol 2003;130:597–600. [PubMed] [Google Scholar]
- Marque M, Vabres M, Prigent F, Guillot B, Bessis D. Congenital melanotic macules of the tongue. Ann Dermatol Venereol 2008;135:567–570. [CrossRef] [PubMed] [Google Scholar]
- Dohil MA, Billman G, pransky S, Guillot B, Bessis D. The congenital lingual melanotic macule. Arch Dermatol 2003;139:767–770. [CrossRef] [PubMed] [Google Scholar]
- Higham P, Alawi F, Stoopler ET. Medical management update: Peutz Jeghers syndrome. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 2010;109:5–11. [CrossRef] [PubMed] [Google Scholar]
- Reychler H. Naevus naevocellulaire buccal. Rev Stomatol Chir Maxillofac 1988;89:97–101. [PubMed] [Google Scholar]
- Buchner A, Hansen LS. Amalgam pigmentation (amalgam tatoo) of the oral mucosa; a clinicopathologic study of 268 cases. Oral Surg 1980;49:139–147. [Google Scholar]
All Figures
 |
Fig. 1 Intraoral photography of an essential melanosis or Laugier–Hunziker syndrome of the attached gingiva at the neck of the teeth 11 and 21. |
| In the text | |
 |
Fig. 2 Histological section of the essential melanosis or Laugier–Hunziker syndrome (HES stain, ×10). Papillomatosis (A), melanin hyperpigmentation of the basal layer (B), and a surface keratosis (C) may be noted. There is no melanocyte proliferation. |
| In the text | |
 |
Fig. 3 Intraoral photography after 3 months of healing indicating the restoration of normalcy. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.


